

Name: Tragacanth
CAS No: Tragacanth gum [9000-65-1]
BP: Tragacanth JP: Tragacanth PhEur: Tragacanth USP-NF: Tragacanth
E413; goat’s thorn; gum benjamin; gum dragon; gum tragacanth; persian tragacanth; trag; tragant; tragacantha.
Tragacanth gum [9000-65-1]
Tragacanth is a naturally occurring dried gum obtained from Astragalus gummifer Labillardie`re and other species of Astragalus grown in Western Asia; The gum consists of a mixture of water-insoluble and watersoluble polysaccharides. Bassorin, which constitutes 60–70% of the gum, is the main water-insoluble portion, while the remainder of the gum consists of the water-soluble material tragacanthin. On hydrolysis, tragacanthin yields L-arabinose, L-fucose, D-xylose, Dgalactose, and D-galacturonic acid. Tragacanth gum also contains small amounts of cellulose, starch, protein, and ash. Tragacanth gum has an approximate molecular weight of 840 000.
Tragacanth gum is used as an emulsifying and suspending agent in a variety of pharmaceutical formulations. It is used in creams, gels, and emulsions at various concentrations according to the application of the formulation and the grade of gum used. Tragacanth gum is also used similarly in cosmetics and food products, and has been used as a diluent in tablet formulations.
Tragacanth gum occurs as flattened, lamellated, frequently curved fragments, or as straight or spirally twisted linear pieces from 0.5–2.5mm in thickness; it may also be obtained in a powdered form. White to yellowish in color, tragacanth is a translucent, odorless substance, with an insipid mucilaginous taste.
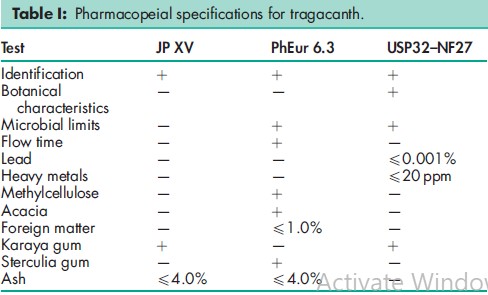
See Table I.
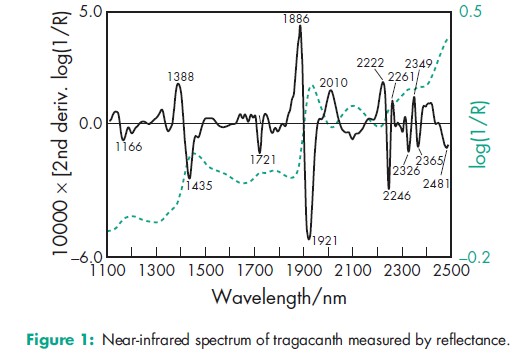
Acidity/alkalinity pH = 5–6 for a 1% w/v aqueous dispersion. Acid value 2–5 Moisture content 415% w/w NIR spectra see Figure 1. Particle size distribution For powdered grades 50% w/w passes through a 73.7 mm mesh. Solubility Practically insoluble in water, ethanol (95%), and other organic solvents. Although insoluble in water, tragacanth gum swells rapidly in 10 times its own weight of either hot or cold water to produce viscous colloidal sols or semigels. Specific gravity 1.250–1.385 Viscosity (dynamic) The viscosity of tragacanth dispersions varies according to the grade and source of the material. Typically, 1% w/v aqueous dispersions may range in viscosity from 100–4000 mPa s (100–4000 cP) at 208C. Viscosity increases with increasing temperature and concentration, and decreases with increasing pH. Maximum initial viscosity occurs at pH 8, although the greatest stability of tragacanth dispersions occurs at about pH 5.
Both the flaked and powdered forms of tragacanth are stable. Tragacanth gels are liable to exhibit microbial contamination with enterobacterial species, and stock solutions should therefore contain suitable antimicrobial preservatives. In emulsions, glycerin or propylene glycol are used as preservatives; in gel formulations, tragacanth is usually preserved with either 0.1% w/v benzoic acid or sodium benzoate. A combination of 0.17% w/v methylparaben and 0.03% w/v propylparaben is also an effective preservative for tragacanth gels;(1) see also Section 12. Gels may be sterilized by autoclaving. Sterilization by gamma irradiation causes a marked reduction in the viscosity of tragacanth dispersions.(2) Tragacanth dispersions are most stable at pH 4–8, although stability is satisfactory at higher pH or as low as pH 2. The bulk material should be stored in an airtight container in a cool, dry place.
At pH 7, tragacanth has been reported to considerably reduce the efficacy of the antimicrobial preservatives benzalkonium chloride, chlorobutanol, and methylparaben, and to a lesser extent that of phenol and phenylmercuric acetate.(3) However, at pH < 5 tragacanth was reported to have no adverse effects on the preservative efficacy of benzoic acid, chlorobutanol, or methylparaben.(1) The addition of strong mineral and organic acids can reduce the viscosity of tragacanth dispersions. Viscosity may also be reduced by the addition of alkali or sodium chloride, particularly if the dispersion is heated. Tragacanth is compatible with relatively high salt concentrations and most other natural and synthetic suspending agents such as acacia, carboxymethylcellulose, starch, and sucrose. A yellow colored, stringy, precipitate is formed with 10% w/v ferric chloride solution.
Tragacanth gum is the air-dried gum obtained from Astragalus gummifer Labillardie`re and other species of Astragalus grown principally in Iran, Syria, and Turkey. A low-quality gum is obtained by collecting the natural air-dried exudate from Astragalus bushes. A higher-grade material is obtained by making incisions in the trunk and branches of the bush, which are held open with variously sized wooden pegs. The exudate is left to drain from the incision and dry naturally in the air before being collected. The size and position of the wooden wedges determine the physical form of the exudate, while the drying conditions determine the color of the gum. After collection, the tragacanth gum is sorted by hand into various grades, such as ribbons or flakes.
Tragacanth has been used for many years in oral pharmaceutical formulations and food products, and is generally regarded as an essentially nontoxic material. Tragacanth has been shown to be noncarcinogenic.(4) However, hypersensitivity reactions, which are occasionaly severe, have been reported following ingestion of products containing tragacanth.(5,6) Contact dermatitis has also been reported following the topical use of tragacanth formulations.( 7) The WHO has not specified an acceptable daily intake for tragacanth gum, as the daily intake necessary to achieve a desired effect, and its background levels in food, were not considered to be a hazard to health.(8) LD50 (hamster, oral): 8.8 g/kg(9) LD50 (mouse, oral): 10 g/kg LD50 (rabbit, oral): 7.2 g/kg LD50 (rat, oral): 16.4 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Tragacanth gum may be irritant to the skin and eyes. Eye protection, gloves, and a dust mask are recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (buccal/ sublingual tablets, oral powders, suspensions, syrups, and tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Tragacanth gum is a naturally occurring material whose physical properties vary greatly according to the grade and source of the material. Samples can contain relatively high levels of bacterial contamination.(10,11) Hog gum (caramania gum), obtained from species of Prunus, and sterculia gum have been used in industrial applications as substitutes for tragacanth. Powdered tragacanth gum tends to form lumps when added to water, and aqueous dispersions should therefore be agitated vigorously with a high-speed mixer. However, aqueous dispersions are more readily prepared by first prewetting the tragacanth with a small quantity of a wetting agent such as ethanol (95%), glycerin, or propylene glycol. If lumps form, they usually disperse on standing. Dispersion is generally complete after 1 hour. If other powders, such as sucrose, are to be incorporated into a tragacanth formulation the powders are best mixed together in the dry state. Some pharmacopeias, such as JP XV, contain a specification for powdered tragacanth.