

Name: Oleic Acid
CAS No: (Z)-9-Octadecenoic acid [112-80-1]
BP: Oleic Acid PhEur: Oleic Acid USP-NF: Oleic Acid
Acidum oleicum; Crodolene; Crossential 094; elaic acid; Emersol; Glycon; Groco; Hy-Phi; Industrene; Metaupon; Neo-Fat; cis-9- octadecenoic acid; 9,10-octadecenoic acid; oleinic acid; Priolene.
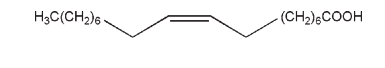
(Z)-9-Octadecenoic acid [112-80-1]
C18H34O2 , 282.47
Oleic acid is used as an emulsifying agent in foods and topical pharmaceutical formulations. It has also been used as a penetration enhancer in transdermal formulations,(1–14) to improve the bioavailability of poorly water-soluble drugs in tablet formulations,(15) and as part of a vehicle in soft gelatin capsules, in topical microemulsion formulations,(16–19) in oral self-emulsifying drug delivery systems,(20,21) in oral mucoadhesive patches,(22) and in a metered dose inhaler.(23) Oleic acid was shown to be an important factor in the hypoglycemic effect produced by multiple emulsions containing insulin intended for intestinal delivery of insulin.(24) The phase behavior of sonicated dispersions of oleic acid has been described,(25) and mechanisms for the topical penetrationenhancing actions of oleic acid have been presented.(26) Oleic acid has been reported to act as an ileal ‘brake’ that slows down the transit of luminal contents through the distal portion of the small bowel.(27) Oleic acid labeled with 131I and 3 H is used in medical imaging.
A yellowish to pale brown, oily liquid with a characteristic lard-like odor and taste. Oleic acid consists chiefly of (Z)-9-octadecenoic acid together with varying amounts of saturated and other unsaturated acids. It may contain a suitable antioxidant.
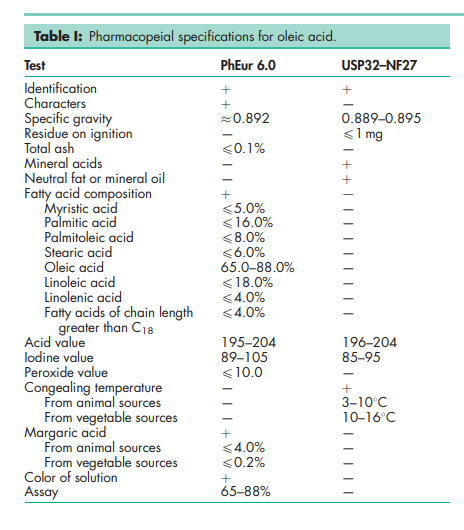
See Table I.
Acidity/alkalinity pH = 4.4 (saturated aqueous solution) Autoignition temperature 3638C Boiling point 2868C at 13.3 kPa (100 mmHg) (decomposition at 80–1008C) Density 0.895 g/cm3 Flash point 1898C Melting point 13–148C;(28) pure oleic acid solidifies at 48C(29) Refractive index nD 26 = 1.4585 Solubility Miscible with benzene, chloroform, ethanol (95%), ether, hexane, and fixed and volatile oils; practically insoluble in water. Vapor pressure 133 Pa (1 mmHg) at 176.58C Viscosity (dynamic) 26 mPa s (26 cP) at 258C
On exposure to air, oleic acid gradually absorbs oxygen, darkens in color, and develops a more pronounced odor. At atmospheric pressure, it decomposes when heated at 80–1008C. Oleic acid should be stored in a well-filled, well-closed container, protected from light, in a cool, dry place.
Incompatible with aluminum, calcium, heavy metals, iodine solutions, perchloric acid, and oxidizing agents. Oleic acid reacts with alkalis to form soaps.
Oleic acid is obtained by the hydrolysis of various animal and vegetable fats or oils, such as olive oil, followed by separation of the liquid acids. It consists chiefly of (Z)-9-octadecenoic acid. Oleic acid that is to be used systemically should be prepared from edible sources.
Oleic acid is used in oral and topical pharmaceutical formulations. In vitro tests have shown that oleic acid causes rupture of red blood cells (hemolysis), and intravenous injection or ingestion of a large quantity of oleic acid can therefore be harmful. The effects of oleic acid on alveolar(30) and buccal(31) epithelial cells in vitro have also been studied; the in vitro and in vivo effects of oleic acid on rat skin have been reported.(32) Oleic acid is a moderate skin irritant; it should not be used in eye preparations. An acceptable daily intake for the calcium, sodium, and potassium salts of oleic acid was not specified by the WHO since the total daily intake of these materials in foods was such that they did not pose a hazard to health.(33) LD50 (mouse, IV): 0.23 g/kg(34) LD50 (rat, IV): 2.4 mg/kg LD50 (rat, oral): 74 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Gloves and eye protection are recommended.
GRAS listed. Included in the FDA Inactive Ingredients Database (inhalation and nasal aerosols, tablets, topical and transdermal preparations). Included in nonparenteral medicines (metered dose inhalers; oral capsules; oral prolonged release granules; topical creams and gels) licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Ethyl oleate.
Several grades of oleic acid are commercially available, ranging in color from pale yellow to reddish brown. Different grades become turbid at varying temperatures depending upon the amount of saturated acid present. Usually, oleic acid contains 7–12% saturated acids, such as stearic and palmitic acid, together with other unsaturated acids, such as linoleic acid. A specification for oleic acid is contained in the Food Chemicals Codex (FCC).(35) The EINECS number for oleic acid is 204-007-1. The PubChem Compound ID (CID) for oleic acid includes 965 and 445639.