

Name: Sodium Metabisulfite
CAS No: Sodium pyrosulfite [7681-57-4]
BP: Sodium Metabisulphite JP: Sodium Pyrosulfite PhEur: Sodium Metabisulphite USP-NF: Sodium Metabisulfite
Disodium disulfite; disodium pyrosulfite; disulfurous acid, disodium salt; E223; natrii disulfis; natrii metabisulfis; sodium acid sulfite.
Sodium pyrosulfite [7681-57-4]
Na2S2O5 190.1 Sodium metabisulfite contains 24.19% sodium, 42.08% oxygen, and 33.73% sulfur.
See Section 4.
Sodium metabisulfite is used as an antioxidant in oral, parenteral, and topical pharmaceutical formulations, at concentrations of 0.01–1.0% w/v, and at a concentration of approximately 27% w/v in intramuscular injection preparations. Primarily, sodium metabisulfite is used in acidic preparations; for alkaline preparations, sodium sulfite is usually preferred; see Section 18. Sodium metabisulfite also has some antimicrobial activity, which is greatest at acid pH, and may be used as a preservative in oral preparations such as syrups. In the food industry and in wine production, sodium metabisulfite is similarly used as an antioxidant, antimicrobial preservative, and antibrowning agent. However, at concentrations above about 550 ppm it imparts a noticeable flavor to preparations. Sodium metabisulfite usually contains small amounts of sodium sulfite and sodium sulfate.
Sodium metabisulfite occurs as colorless, prismatic crystals or as a white to creamy-white crystalline powder that has the odor of sulfur dioxide and an acidic, saline taste. Sodium metabisulfite crystallizes from cold water as a hydrate containing seven water molecules
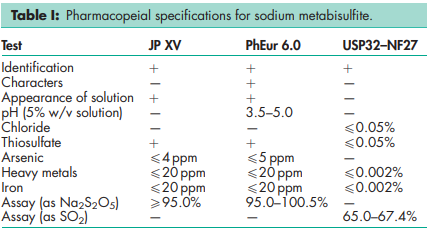
See Table I.
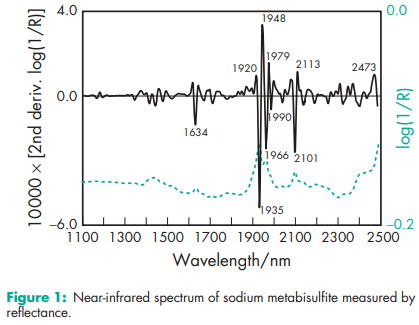
Acidity/alkalinity pH = 3.5–5.0 for a 5% w/v aqueous solution at 208C. Melting point Sodium metabisulfite melts with decomposition at less than 1508C. NIR spectra see Figure 1. Osmolarity A 1.38% w/v aqueous solution is isoosmotic with serum. Solubility

On exposure to air and moisture, sodium metabisulfite is slowly oxidized to sodium sulfate with disintegration of the crystals.(1) Addition of strong acids to the solid liberates sulfur dioxide. In water, sodium metabisulfite is immediately converted to sodium (Naþ) and bisulfite (HSO3 ) ions. Aqueous sodium metabisulfite solutions also decompose in air, especially on heating. Solutions that are to be sterilized by autoclaving should be filled into containers in which the air has been replaced with an inert gas, such as nitrogen. The addition of dextrose to aqueous sodium metabisulfite solutions results in a decrease in the stability of the metabisulfite.(2) The bulk material should be stored in a well-closed container, protected from light, in a cool, dry place.
Sodium metabisulfite reacts with sympathomimetics and other drugs that are ortho- or para-hydroxybenzyl alcohol derivatives to form sulfonic acid derivatives possessing little or no pharmacological activity. The most important drugs subject to this inactivation are epinephrine (adrenaline) and its derivatives.(3) In addition, sodium metabisulfite is incompatible with chloramphenicol owing to a more complex reaction;(3) it also inactivates cisplatin in solution.(4,5) It is incompatible with phenylmercuric acetate when autoclaved in eye drop preparations.(6) Sodium metabisulfite may react with the rubber caps of multidose vials, which should therefore be pretreated with sodium metabisulfite solution.(7)
Sodium metabisulfite is prepared by saturating a solution of sodium hydroxide with sulfur dioxide and allowing crystallization to occur; hydrogen is passed through the solution to exclude air. Sodium metabisulfite may also be prepared by saturating a solution of sodium carbonate with sulfur dioxide and allowing crystallization to occur, or by thermally dehydrating sodium bisulfite.
Sodium metabisulfite is widely used as an antioxidant in oral, topical, and parenteral pharmaceutical formulations; it is also widely used in food products. Although it is extensively used in a variety of preparations, sodium metabisulfite and other sulfites have been associated with a number of severe to fatal adverse reactions.(8–19) These are usually hypersensitivity-type reactions and include bronchospasm and anaphylaxis. Allergy to sulfite antioxidants is estimated to occur in 5–10% of asthmatics, although adverse reactions may also occur in nonasthmatics with no history of allergy. Following oral ingestion, sodium metabisulfite is oxidized to sulfate and is excreted in urine. Ingestion may result in gastric irritation, owing to the liberation of sulfurous acid, while ingestion of large amounts of sodium metabisulfite can cause colic, diarrhea, circulatory disturbances, CNS depression, and death. In Europe, the acceptable daily intake of sodium metabisulfite and other sulfites used in foodstuffs has been set at up to 3.5 mg/kg body-weight, calculated as sulfur dioxide (SO2). The WHO has similarly also set an acceptable daily intake of sodium metabisulfite, and other sulfites, at up to 7.0 mg/kg body-weight, calculated as sulfur dioxide (SO2).(20) LD50 (rat, IV): 0.12 g/kg(21)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Sodium metabisulfite may be irritant to the skin and eyes; eye protection and gloves are recommended. In the UK, the long-term (8-hour TWA) workplace exposure limit for sodium metabisulfite is 5 mg/m3 . (22)
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (epidural;inhalation; IM and IV injections; ophthalmic solutions; oral preparations; rectal, topical, and vaginal preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Potassium metabisulfite; sodium bisulfite; sodium sulfite. Sodium bisulfite Empirical formula NaHSO3 Molecular weight 104.07 CAS number [7631-90-5] Synonyms E222; sodium hydrogen sulfite. Appearance White crystalline powder. Density 1.48 g/cm3 Solubility Soluble 1 in 3.5 parts of water at 208C; 1 in 2 parts of water at 1008C; and 1 in 70 parts of ethanol (95%). Freely soluble in glycerol. Aqueous solution is acidic. Comments Most substances sold as sodium bisulfite contain significant, variable, amounts of sodium metabisulfite, as the latter is less hygroscopic and more stable during storage and shipmen
Sodium metabisulfite is used as an antioxidant at low pH, sodium bisulfite at intermediate pH, and sodium sulfite at higher pH values. A specification for sodium metabisulfite is contained in the Food Chemicals Codex (FCC).(23) The EINECS number for sodium metabisulfite is 231-673-0.