

Name: Triethanolamine
CAS No: 2,20,200-Nitrilotriethanol [102-71-6]
BP: Triethanolamine PhEur: Trolamine USP-NF: Trolamine
TEA; Tealan; triethylolamine; trihydroxytriethylamine; tris (hydroxyethyl)amine; trolaminum.
2,20,200-Nitrilotriethanol [102-71-6]
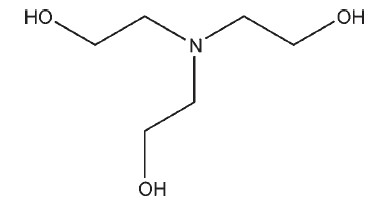
C6H15NO3 149.19
Triethanolamine is widely used in topical pharmaceutical formulations, primarily in the formation of emulsions. When mixed in equimolar proportions with a fatty acid, such as stearic acid or oleic acid, triethanolamine forms an anionic soap with a pH of about 8, which may be used as an emulsifying agent to produce fine-grained, stable oil-in-water emulsions. Concentrations that are typically used for emulsification are 2–4% v/v of triethanolamine and 2–5 times that of fatty acids. In the case of mineral oils, 5% v/v of triethanolamine will be needed, with an appropriate increase in the amount of fatty acid used. Preparations that contain triethanolamine soaps tend to darken on storage. However, discoloration may be reduced by avoiding exposure to light and contact with metals and metal ions. Triethanolamine is also used in salt formation for injectable solutions and in topical analgesic preparations. It is also used in sun screen preparations.(1) Triethanolamine is used as an intermediate in the manufacturing of surfactants, textile specialties, waxes, polishes, herbicides, petroleum demulsifiers, toilet goods, cement additives, and cutting oils. Triethanolamine is also claimed to be used for the production of lubricants for the rubber gloves and textile industries. Other general uses are as buffers, solvents, and polymer plasticizers, and as a humectant.
Triethanolamine is a clear, colorless to pale yellow-colored viscous liquid having a slight ammoniacal odor. It is a mixture of bases, mainly 2,20,200-nitrilotriethanol, although it also contains 2,20- iminobisethanol (diethanolamine) and smaller amounts of 2- aminoethanol (monoethanolamine).
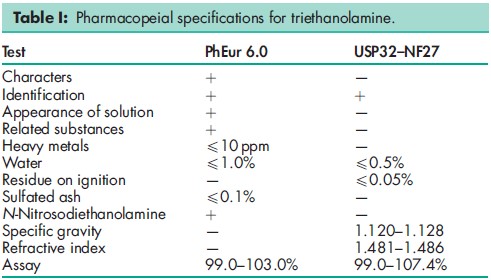
See Table I.
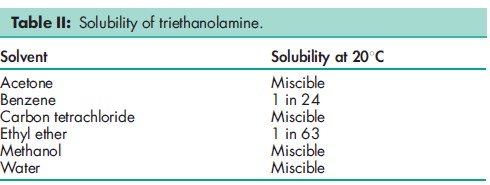
Acidity/alkalinity pH = 10.5 (0.1N solution) Boiling point 3358C Flash point 2088C Freezing point 21.68C Hygroscopicity Very hygroscopic. Melting point 20–218C Moisture content 0.09% Solubility see Table II. Surface tension 48.9mN/m (48.9 dynes/cm) at 258C Viscosity (dynamic) 590 mPa s (590 cP) at 308C
Triethanolamine may turn brown on exposure to air and light. The 85% grade of triethanolamine tends to stratify below 158C; homegeneity can be restored by warming and mixing before use. Triethanolamine should be stored in an airtight container protected from light, in a cool, dry place. See Monoethanolamine for further information.
Triethanolamine is a tertiary amine that contains hydroxy groups; it is capable of undergoing reactions typical of tertiary amines and alcohols. Triethanolamine will react with mineral acids to form crystalline salts and esters. With the higher fatty acids, triethanolamine forms salts that are soluble in water and have characteristics of soaps. Triethanolamine will also react with copper to form complex salts. Discoloration and precipitation can take place in the presence of heavy metal salts. Triethanolamine can react with reagents such as thionyl chloride to replace the hydroxy groups with halogens. The products of these reactions are very toxic, resembling other nitrogen mustards.
Triethanolamine is prepared commercially by the ammonolysis of ethylene oxide. The reaction yields a mixture of monoethanolamine, diethanolamine, and triethanolamine, which are separated to obtain the pure products.
Triethanolamine is used primarily as an emulsifying agent in a variety of topical pharmaceutical preparations. Although generally regarded as a nontoxic material,(2) triethanolamine may cause hypersensitivity or be irritant to the skin when present in formulated products. The lethal human oral dose of triethanolamine is estimated to be 5–15 g/kg body-weight. Following concern about the possible production of nitrosamines in the stomach, the Swiss authorities have restricted the use of triethanolamine to preparations intended for external use.(3) LD50 (guinea pig, oral): 5.3 g/kg(4) LD50 (mouse, IP): 1.45 g/kg LD50 (mouse, oral): 7.4 g/kg LD50 (rat, oral): 8 g/kg
Triethanolamine may be irritant to the skin, eyes, and mucous membranes. Inhalation of vapor may be harmful. Protective clothing, gloves, eye protection, and a respirator are recommended. Ideally, triethanolamine should be handled in a fume cupboard. On heating, triethanolamine forms highly toxic nitrous fumes. Triethanolamine is combustible.
Included in the FDA Inactive Ingredients Database (rectal, topical, and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Diethanolamine; monoethanolamine.
Various grades of triethanolamine are available. The standard commercial grade contains 85% triethanolamine. The superior grade contains 98–99% triethanolamine. One volume part of triethanolamine with 5–7 parts of a mixture of CaO2 and ZnO2 is used as a filling material that enhances the restorative process in periodontal tissues. Triethanolamine is recommended as the preferred stabilizer to be used in latex polymerization because of its weak mutagenic effect in the Ames tests. The EINECS number for triethanolamine is 203-049-8.