

Name: Nitrogen
CAS No: Nitrogen [7727-37-9]
BP: Nitrogen JP: Nitrogen PhEur: Nitrogen USP-NF: Nitrogen
Azote; E941; nitrogenium.
Nitrogen [7727-37-9]
N2, 28.01
Nitrogen and other compressed gases such as carbon dioxide and nitrous oxide are used as propellants for topical pharmaceutical aerosols. They are also used in other aerosol products that work satisfactorily with the coarse aerosol spray produced with compressed gases, e.g. furniture polish and window cleaner. Nitrogen is insoluble in water and other solvents, and therefore remains separated from the actual pharmaceutical formulation. Advantages of compressed gases as aerosol propellants are that they are less expensive; of low toxicity; and practically odorless and tasteless. In contrast to liquefied gases, their pressures change relatively little with temperature. However, there is no reservoir of propellant in the aerosol and as a result the pressure decreases as the product is used, changing the spray characteristics. Misuse of a product by the consumer, such as using a product inverted, results in the discharge of the vapor phase instead of the liquid phase. Most of the propellant is contained in the vapor phase and therefore some of the propellant will be lost and the spray characteristics will be altered. Additionally, the sprays produced using compressed gases are very wet. However, recent developments in valve technology have reduced the risk of misuse by making available valves which will spray only the product (not propellant) regardless of the position of the container. Additionally, barrier systems will also prevent loss of propellant, and have been used for pharmaceuticals and cosmetic aerosol sprays and foams utilizing nitrogen as the propellant. Nitrogen is also used to displace air from solutions subject to oxidation, by sparging, and to replace air in the headspace above products in their final packaging, e.g. in parenteral products packaged in glass ampoules. Nitrogen is also used for the same purpose in many food products.
Nitrogen occurs naturally as approximately 78% v/v of the atmosphere. It is a nonreactive, noncombustible, colorless, tasteless, and odorless gas. It is usually handled as a compressed gas, stored in metal cylinders.
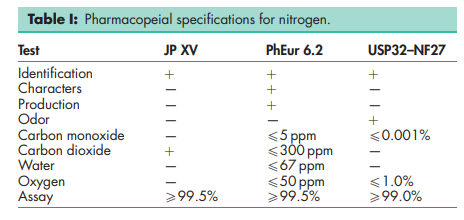
See Table I.
Density 0.967 g/cm3 for vapor at 218C. Flammability Nonflammable Solubility Practically insoluble in water and most solvents; soluble in water under pressure. Vapor density (absolute) 1.25 g/cm3 at standard temperature and pressure. Vapor density (relative) 0.97 (air = 1)
Nitrogen is stable and chemically unreactive. It should be stored in tightly sealed metal cylinders in a cool, dry place.
Generally compatible with most materials encountered in pharmaceutical formulations and food products
Nitrogen is obtained commercially, in large quantities, by the fractional distillation of liquefied air.
Nitrogen is generally regarded as a nontoxic and nonirritant material. However, it is an asphyxiant and inhalation of large quantities is therefore hazardous.
Handle in accordance with procedures for handling metal cylinders containing liquefied or compressed gases. Eye protection, gloves, and protective clothing are recommended. Nitrogen is an asphyxiant and should be handled in a well-ventilated environment.
GRAS listed. Included in the FDA Inactive Ingredients Database (injections; dental preparations; nasal sprays; oral solutions; rectal gels). Accepted for use as a food additive in Europe. Included in parenteral and nonparenteral medicines licensed in the UK and USA. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Carbon dioxide; nitrous oxide.
Different grades of nitrogen are commercially available that have, for example, especially low moisture levels. Nitrogen is commonly used as a component of the gas mixtures breathed by divers. Under high pressure, such as when diving at great depths, nitrogen will dissolve in blood and lipid. If decompression is too rapid, decompression sickness may occur when the nitrogen effervesces from body stores to form gas emboli. A specification for nitrogen is contained in the Food Chemicals Codex (FCC).(1) The EINECS number for nitrogen is 231-783-9. The PubChem Compound ID (CID) for nitrogen is 947.