

Name: Nitrous Oxide
CAS No: Dinitrogen oxide [10024-97-2]
BP: Nitrous Oxide JP: Nitrous Oxide PhEur: Nitrous Oxide USP: Nitrous Oxide
Dinitrogenii oxidum; dinitrogen monoxide; E942; laughing gas; nitrogen monoxide.
Dinitrogen oxide [10024-97-2]
N2O ,44.01
Nitrous oxide and other compressed gases such as carbon dioxide and nitrogen are used as propellants for topical pharmaceutical aerosols. They are also used in other aerosol products that work satisfactorily with the coarse aerosol spray that is produced with compressed gases, e.g. furniture polish and window cleaner. The advantages of compressed gases as aerosol propellants are that they are less expensive, of low toxicity, and practically odorless and tasteless. In contrast to liquefied gases, their pressures change relatively little with temperature. However, there is no reservoir of propellant in the aerosol, and as a result the pressure decreases as the product is used, changing the spray characteristics. Misuse of a product by the consumer, such as using a product inverted, results in the discharge of the vapor phase instead of the liquid phase. Since most of the propellant is contained in the vapor phase, some of the propellant will be lost and the spray characteristics will be altered. Additionally, the sprays produced using compressed gases are very wet. However, recent developments in valve technology have reduced the risk of misuse by making available valves which will spray only the product (not propellant) regardless of the position of the container. Additionally, barrier systems will also prevent loss of propellant, and have found increased use with this propellant. Therapeutically, nitrous oxide is best known as an anesthetic administered by inhalation. When used as an anesthetic it has strong analgesic properties but produces little muscle relaxation. Nitrous oxide is always administered in conjunction with oxygen since on its own it is hypoxic.
Nitrous oxide is a nonflammable, colorless and odorless, sweettasting gas. It is usually handled as a compressed gas, stored in metal cylinders.
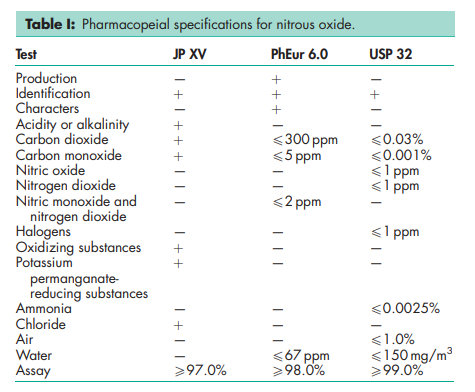
See Table I.
Density 1.53 g/cm3 Flammability Nonflammable, but supports combustion. Solubility Freely soluble in chloroform, ethanol (95%), ether, and oils; soluble 1 in 1.5 volumes of water at 208C and 101.3 kPa pressure. Vapor density (absolute) 1.97 g/cm3 at standard temperature and pressure. Vapor density (relative) 1.52 (air = 1)
Nitrous oxide is essentially nonreactive and stable except at high temperatures; at a temperature greater than 5008C nitrous oxide decomposes to nitrogen and oxygen. Explosive mixtures may be formed with other gases such as ammonia, hydrogen, and other fuels. Nitrous oxide should be stored in a tightly sealed metal cylinder in a cool, dry place.
Nitrous oxide is generally compatible with most materials encountered in pharmaceutical formulations, although it may react as a mild oxidizing agent.
Nitrous oxide is prepared by heating ammonium nitrate to about 1708C. This reaction also forms water.
Nitrous oxide is most commonly used therapeutically as an anesthetic and analgesic. Reports of adverse reactions to nitrous oxide therefore generally concern its therapeutic use, where relatively large quantities of the gas may be inhaled, rather than its use as an excipient. The main complications associated with nitrous oxide inhalation occur as a result of hypoxia. Prolonged administration may also be harmful. Nitrous oxide is rapidly absorbed on inhalation.
Handle in accordance with procedures for handling metal cylinders containing liquefied or compressed gases. Eye protection, gloves, and protective clothing are recommended. Nitrous oxide is an anesthetic gas and should be handled in a well-ventilated environment. In the UK, the recommended long-term (8-hour TWA) workplace exposure limit for nitrous oxide is 183 mg/m3 (100 ppm).(1)
GRAS listed. Accepted for use as a food additive in Europe. Included in nonparenteral medicines licensed in the UK and USA. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Carbon dioxide; nitrogen.
A mixture of 50% nitrous oxide and 50% oxygen ( Entonox, BOC) is commonly used as an analgesic administered by inhalation. A specification for nitrous oxide is contained in the Food Chemicals Codex (FCC).(2) The EINECS number for nitrous oxide is 233-032-0. The PubChem Compound ID (CID) for nitrous oxide is 948.