

Name: Aluminum Hydroxide Adjuvant
CAS No: Aluminum oxyhydroxide [21645-51-2]
PhEur: Aluminium Hydroxide, Hydrated, for Adsorption
Alhydrogel; aluminii hydroxidum hydricum ad adsorptionem; aluminium hydroxide adjuvant; aluminium oxyhydroxide; poorly crystalline boehmite; pseudoboehmite; Rehydragel.
Aluminum oxyhydroxide [21645-51-2]
AlO(OH) 59.99
Structural hydroxyl groups form hydrogen bonds between AlO(OH) octahedral sheets, where hydroxyl groups are exposed at the surface. The surface hydroxyl groups produce a pHdependent surface charge by accepting a proton to produce a positive site, or donating a proton to produce a negative site. The pH-dependent surface charge is characterized by the point of zero charge, which is equivalent to the isoelectric point in protein chemistry. The surface hydroxyl groups may also undergo ligand exchange with fluoride, phosphate, carbonate, sulfate, or borate anions.
Aluminum hydroxide adjuvant is used in parenteral human and veterinary vaccines.(1) It activates Th2 immune responses, including IgG and IgE antibody responses. It is also used for the isolation of certain serum components such as blood clotting factors.(2
Aluminum hydroxide adjuvant is a white hydrogel that sediments slowly and forms a clear supernatant.
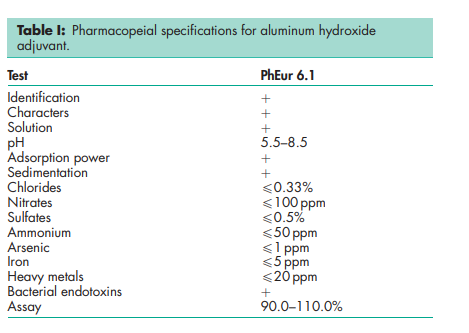
See Table I. Note that the USP 32 includes a monograph for aluminum hydroxide gel, which is a form of aluminum hydroxide that is used as an antacid, in which there is a partial substitution of carbonate for hydroxide. See Section 17.
Acidity/alkalinity pH = 5.5–8.5 Particle size distribution Primary particles are fibrous with average dimensions of 4.5 2.2 10 nm. The primary particles form aggregates of 1–10 mm. Point of zero charge pH = 11.4 Protein binding capacity >0.5 mg BSA/mg equivalent Al2O3 Solubility Soluble in alkali hydroxides and mineral acids. Heat may be required to dissolve the aluminum hydroxide adjuvant. Specific surface area 500 m2 /g.(3) X-ray diffractogram Exhibits characteristic x-ray diffraction pattern having diffraction bands at 6.46, 3.18, 2.35, 1.86, 1.44 and 1.31 A˚ .
Aluminum hydroxide adjuvant is stable for at least 2 years when stored at 4–308C in well-sealed inert containers. It must not be allowed to freeze as the hydrated colloid structure will be irreversibly damaged.
When exposed to phosphate, carbonate, sulfate, or borate anions, the point of zero charge for aluminum hydroxide adjuvant decreases.
Aluminum hydroxide adjuvant is prepared by the precipitation of a soluble aluminum salt by an alkali hydroxide, or the precipitation of an alkali aluminate by acid.
Aluminum hydroxide adjuvant is intended for use in parenteral vaccines and is generally regarded as nontoxic. It may cause mild irritation, dryness, and dermatitis on skin contact. On eye contact, aluminum hydroxide adjuvant may also cause redness, conjunctivitis, and short-term mild irritation. Ingestion of large amounts may cause gastrointestinal irritation with nausea, vomiting, and constipation. Inhalation of the dried product may cause respiratory irritation and cough. Type I hypersensitivity reactions following parenteral administration have been reported.(4)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended.
GRAS listed. Accepted for use in human and veterinary parenteral vaccines in Europe and the USA. The limits for use in human vaccines are 0.85 mg aluminum/dose (FDA) and 1.25 mg aluminum/dose (WHO). There are no established limits for use in veterinary vaccines. Reported in the EPA TSCA Inventory.
Aluminum phosphate adjuvant
Different grades of aluminum hydroxide adjuvant with various concentrations, protein binding capacities, and points of zero charge are available. The impurity limits at 2% equivalent Al2O3 are Cl < 0.5%; SO4 < 0.5%; PO4 < 0.1%; NO3 < 0.1%; NH4 < 0.1%; Fe < 20 ppm; As < 0.6 ppm; and heavy metals < 20 ppm. The aluminum hydroxide gel referred to in the USP 32 is used in cosmetics as an emollient, filler, humectant, a mild astringent, and viscosity controlling agent. In pharmaceutical preparations it is used as an adsorbent, and as a protein binder.(5) It is also used therapeutically as an antacid, and as an abrasive in dentrifrices. It is not, however, used as a vaccine adjuvant.