

Name: Propylparaben Sodium
CAS No: Sodium 4-propoxycarbonylphenolate [35285-69-9]
BP: Sodium Propyl Hydroxybenzoate PhEur: Sodium Propyl Parahydroxybenzoate USP-NF: Propylparaben Sodium
E217; 4-hydroxybenzoic acid propyl ester, sodium salt; Nipasol M Sodium; parasept; propyl 4-hydroxybenzoate, sodium salt; propyl p-hydroxybenzoate, sodium salt; propylis parahydroxybenzoas natricus; sodium 4-propoxycarbonylphenolate; sodium propyl phydroxybenzoate; soluble propyl hydroxybenzoate.
Sodium 4-propoxycarbonylphenolate [35285-69-9]
C10H11NaO3, 202.2
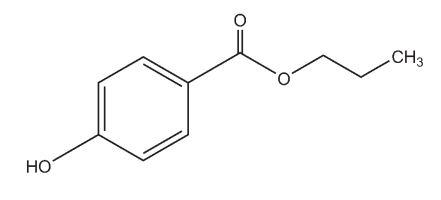
Propylparaben sodium is used as an antimicrobial or antifungal preservative in oral pharmaceuticals and in many water-based cosmetics. It is generally used in combination with other paraben esters
Propylparaben sodium occurs as a white, crystalline, odorless or almost odorless powder.
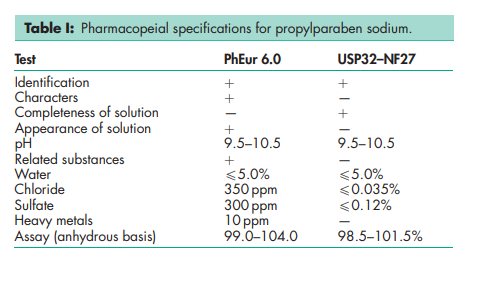
See Table I.
Acidity/alkalinity pH = 9.5–10.5 (0.1% w/v aqueous solution) Dissociation constant pKa = 8.4 at 228C Partition coefficient log P (octanol : water) = 3.0 Solubility see Table II.

Propylparaben sodium is stable under normal conditions. It decomposes on heating. Store in a tightly closed container.
The activity of propylparaben sodium can be adversely affected by the presence of other excipients or active ingredients, such as atropine, essential oils, iron, magnesium trisilicate, talc, polysorbate 80 and other nonionic surfactants, sorbitol, weak alkalis, and strong acids.(1)
Propylparaben sodium is produced from benzoic acid
Propylparaben sodium is used in oral pharmaceuticals and cosmetics. The pure form is toxic by the IV route and moderately toxic by ingestion and the IP route. Propylparaben sodium may cause asthma, rashes, and hyperactivity. LD50 (mouse, IP): 0.49 g/kg LD50 (mouse, IV): 0.18 g/kg LD50 (mouse, oral): 3.7 g/kg(2)
Observe normal precautions appropriate to the circumstances and quantity of the material handled. Avoid inhalation or contact with eyes, skin, and clothing. Avoid prolonged or repeated exposure. Wear suitable protective clothing, gloves, eye/face protection, and a respirator.
Included in the FDA Inactive Ingredients Database (oral capsules, tablets, suspensions). Accepted for use as a food additive in Europe. Included in nonparenteral medicines (oral capsules, mixtures, orodispersible tablets, solutions and suspensions; cutaneous emulsions) licensed in the UK.
Butylparaben; ethylparaben; methylparaben; propylparaben.
Propylparaben sodium may be used instead of propylparaben because of its greater aqueous solubility. However, it may cause the pH of a formulation to become more alkaline. The EINECS number for propylparaben sodium is 252-488-1. The PubChem Compound ID (CID) for propylparaben sodium is 23679044.