

Name: Sodium Starch Glycolate
CAS No: Sodium carboxymethyl starch [9063-38-1]
BP: Sodium Starch Glycolate PhEur: Sodium Starch Glycolate USP-NF: Sodium Starch Glycolate
Carboxymethyl starch, sodium salt; carboxymethylamylum natricum; Explosol; Explotab; Glycolys; Primojel; starch carboxymethyl ether, sodium salt; Tablo; Vivastar P.
Sodium carboxymethyl starch [9063-38-1]
The USP32–NF27 describes two types of sodium starch glycolate, Type A and Type B, and states that sodium starch glycolate is the sodium salt of a carboxymethyl ether of starch or of a crosslinked carboxymethyl ether of starch. The PhEur 6.0 describes three types of material: Type A and Type B are described as the sodium salt of a crosslinked partly Ocarboxymethylated potato starch. Type C is described as the sodium salt of a partly O-carboxymethylated starch, crosslinked by physical dehydration. Types A, B, and C are differentiated by their pH, sodium, and sodium chloride content. The PhEur and USP–NF monographs have been harmonized for Type A and Type B variants. Sodium starch glycolate may be characterized by the degree of substitution and crosslinking. The molecular weight is typically 5 105 –1 106 .
Sodium starch glycolate is widely used in oral pharmaceuticals as a disintegrant in capsule(1–6) and tablet formulations.(7–10) It is commonly used in tablets prepared by either direct-compression(11–13) or wet-granulation processes.(14–16) The usual concentration employed in a formulation is between 2% and 8%, with the optimum concentration about 4%, although in many cases 2% is sufficient. Disintegration occurs by rapid uptake of water followed by rapid and enormous swelling.(17–20) Although the effectiveness of many disintegrants is affected by the presence of hydrophobic excipients such as lubricants, the disintegrant efficiency of sodium starch glycolate is unimpaired. Increasing the tablet compression pressure also appears to have no effect on disintegration time.(10–12) Sodium starch glycolate has also been investigated for use as a suspending vehicle.(21)
Sodium starch glycolate is a white or almost white free-flowing very hygroscopic powder. The PhEur 6.0 states that when examined under a microscope it is seen to consist of: granules, irregularly shaped, ovoid or pear-shaped, 30–100 mm in size, or rounded, 10–35 mm in size; compound granules consisting of 2–4 components occur occasionally; the granules have an eccentric hilum and clearly visible concentric striations. Between crossed nicol prisms, the granules show a distinct black cross intersecting at the hilum; small crystals are visible at the surface of the granules. The granules show considerable swelling in contact with water
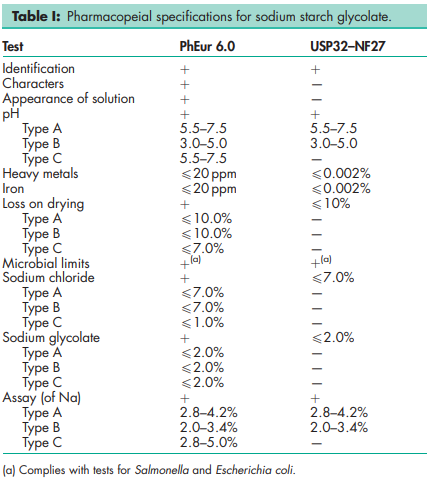
See Table I.
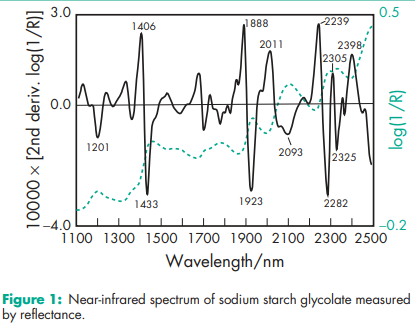
Acidity/alkalinity See Section 9. Density (bulk) 0.756 g/cm3 for Glycolys; 0.81 g/cm3 for Primojel; 0.67 g/cm3 for Tablo. Density (tapped) 0.945 g/cm3 for Glycolys; 0.98 g/cm3 for Primojel; 0.83 g/cm3 for Tablo. Density (true) 1.56 g/cm3 for Primojel; 1.49 g/cm3 for Tablo. Melting point Does not melt, but chars at approximately 2008C. NIR spectra see Figure 1. Particle size distribution 100% of particles less than 106 mm in size. Average particle size (d50) is 38 mm and 42 mm for Primojel by microscopy and sieving, respectively. Solubility Practically insoluble in methylene chloride. It gives a translucent suspension in water. Specific surface area 0.24 m2 /g for Glycolys; 0.185 m2 /g for Primojel; 0.335 m2 /g for Tablo. Swelling capacity In water, sodium starch glycolate swells to up to 300 times its volume. Viscosity (dynamic) 4200 mPa s (200 cP) for a 4% w/v aqueous dispersion; viscosity is 4.26 mPa s for a 2% w/v aqueous dispersion (depending on source and grade). Swelling capacity In water, sodium starch glycolate swells to up to 300 times its volume. Viscosity (dynamic) 4200 mPa s (200 cP) for a 4% w/v aqueous dispersion; viscosity is 4.26 mPa s for a 2% w/v aqueous dispersion (depending on source and grade).




Tablets prepared with sodium starch glycolate have good storage properties.(22–24) Sodium starch glycolate is stable although very hygroscopic, and should be stored in a well-closed container in order to protect it from wide variations of humidity and temperature, which may cause caking. The physical properties of sodium starch glycolate remain unchanged for up to 3 years if it is stored at moderate temperatures and humidity
Sodium starch glycolate is incompatible with ascorbic acid.(25)
Sodium starch glycolate is a substituted derivative of potato starch. Typically, commercial products are also crosslinked using either sodium trimetaphosphate (Types A and B) or dehydration (Type C).(26) Starch is carboxymethylated by reacting it with sodium chloroacetate in an alkaline, nonaqueous medium, typically denatured ethanol or methanol, followed by neutralization with citric acid, acetic acid, or some other acid. Vivastar P is manufactured in methanolic medium, and Explotab in ethanolic medium.
Sodium starch glycolate is widely used in oral pharmaceutical formulations and is generally regarded as a nontoxic and nonirritant material. However, oral ingestion of large quantities may be harmful.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Sodium starch glycolate may be irritant to the eyes; eye protection and gloves are recommended. A dust mask or respirator is recommended for processes that generate a large quantity of dust.
Included in the FDA Inactive Ingredients Database (oral capsules and tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Pregelatinized starch; starch.
Sodium starch glycolate is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. The physical properties of sodium starch glycolate, and hence its effectiveness as a disintegrant, are affected by the degree of crosslinkage, extent of carboxymethylation, and purity.(27,28) Sodium starch glycolate has been reported to interact with glycopeptide antibiotics,(29,30) basic drugs, and increase the photostability of norfloxacin.(31) The solubility of the formulation matrix and mode of incorporation in wet granulation can affect the disintegration time; disintegration times can be slower in tablets containing high levels of soluble excipients.(32) Commercially, sodium starch glycolate is available in a number of speciality grades, e.g. low pH (Explotab Low pH, Glycolys Low pH); low viscosity (Explotab CLV, Glycolys LV); low solvent (Vivastar PSF); and low moisture Glycolys LM. A specification for sodium starch glycolate is included in the Japanese Pharmaceutical Excipients (JPE).(33)