

Name: Pectin
CAS No: Pectin [9000-65-5]
USP: Pectin
Citrus pectin; E440; Genu; methopectin; methyl pectin; methyl pectinate; mexpectin; pectina; pectinic acid.
Pectin [9000-65-5]
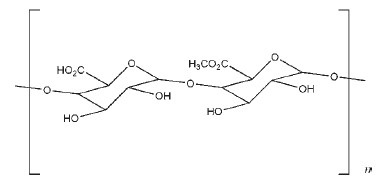
Pectin is a high-molecular-weight, carbohydrate-like plant constituent consisting primarily of chains of galacturonic acid units linked as 1,4-a-glucosides, with a molecular weight of 30 000–100 000.
Pectin is a complex polysaccharide comprising mainly esterified Dgalacturonic acid residues in an a-(1–4) chain. The acid groups along the chain are largely esterified with methoxy groups in the natural product. The hydroxyl groups may also be acetylated. Pectin gelation characteristics can be divided into two types: high-methoxy and low-methoxy gelation, and sometimes the lowmethoxy pectins may contain amine groups. Gelation of highmethoxy pectin usually occurs at pH < 3.5. Low-methoxy pectin is gelled with calcium ions and is not dependent on the presence of acid or high solids content. Amidation may interfere with gelation, causing the process to be delayed. However, gels from amidated pectins have the ability to re-heal after shearing.(1) The USP 32 describes pectin as a purified carbohydrate product obtained from the dilute acid extract of the inner portion of the rind of citrus fruits or from apple pomace. It consists chiefly of partially methoxylated polygalacturonic acids.
Pectin has been used as an adsorbent and bulk-forming agent, and is present in multi-ingredient preparations for the management of diarrhea, constipation, and obesity;(2) it has also been used as an emulsion stabilizer.(3) Experimentally, pectin has been used in gel formulations for the oral sustained delivery of ambroxol.(4) Pectin gel beads have been shown to be an effective medium for controlling the release of a drug within the gastrointestinal (GI) tract.(5) It has also been used in a colon-biodegradable pectin matrix with a pH-sensitive polymeric coating, which retards the onset of drug release, overcoming the problems of pectin solubility in the upper GI tract.(6–9) Amidated pectin matrix patches have been investigated for the transdermal delivery of chloroquine,(10) and gelling pectin formulations for the oral sustained delivery of paracetamol have been investigated in situ.(11) Pectin-based matrices with varying degrees of esterification have been evaluated as oral controlled-release tablets. Lowmethoxy pectins were shown to have a release rate more sensitive to the calcium content of the formulation.(12) Pectins have been used as a component in the preparation of mixed polymer microsphere systems with the intention of producing controlled drug release.(13)
Pectin occurs as a coarse or fine, yellowish-white, odorless powder that has a mucilaginous taste.
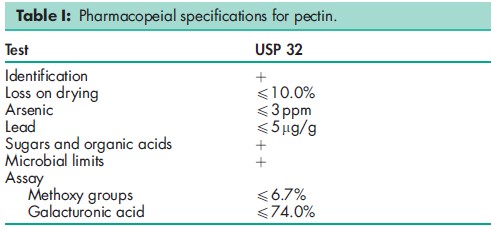
See Table I.
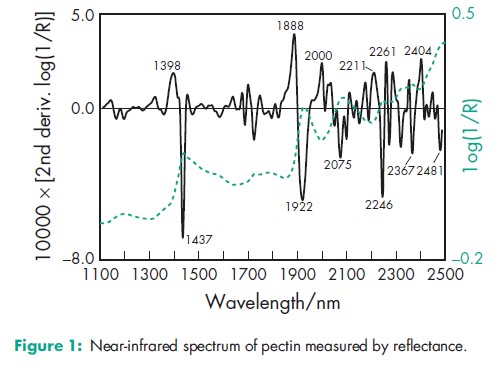
Acidity/alkalinity pH = 6.0–7.2 NIR spectra see Figure 1. Solubility Soluble in water; insoluble in ethanol (95%) and other organic solvents.
Pectin is a nonreactive and stable material; it should be stored in a cool, dry place.
—
Pectin is obtained from the diluted acid extract from the inner portion of the rind of citrus fruits or from apple pomace.
Pectin is used in oral pharmaceutical formulations and food products, and is generally regarded as an essentially nontoxic and nonirritant material. Low toxicity by the subcutaneous route has been reported.(14) LD50 (mouse, SC): 6.4 g/kg(14)
Observe normal precautions appropriate to the circumstances and quantity of material handled. When pectin is heated to decomposition, acrid smoke and irritating fumes are emitted.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (dental paste; oral powders; topical pastes). Included in the Canadian List of Acceptable Non-medicinal Ingredients. Included in nonparenteral medicines licensed in the UK.
—
Pectin has been used in film-coating formulations containing chitosan and hydroxypropylmethyl cellulose in the investigation of the biphasic drug-release properties of film-coated paracetamol tablets, both in vitro,(15,16) and in vivo.(17) It has been shown that chitosan acts as a crosslinking agent for concentrated pectin solutions.(18) Pectin gel systems have been used to show the partition and release of aroma compounds in foods during storage.(19) A specification for pectins is included in the Food Chemical Codex (FCC).(20) In the food industry it is used as an emulsifying agent, gelling agent, thickener, and stabilizer. Cosmetically, it is used as a binder, emulsifying agent and viscosity-controlling agent. The EINECS number for pectin is 232-553-0.