

Name: Sodium Sulfite
CAS No: Sodium sulfite [7757-83-7]
BP: Anhydrous Sodium Sulphite JP: Dried Sodium Sulfite PhEur: Sodium Sulphite, Anhydrous USP-NF: Sodium Sulfite
Disodium sulfite; exsiccated sodium sulfite; E221; natrii sulfis anhydricus; sulfurous acid disodium salt.
Sodium sulfite [7757-83-7]
Na2SO3 126.04
See Section 4.
Sodium sulfite is used as an antioxidant in applications similar to those for sodium metabisulfite.(1) It is also an effective antimicrobial preservative, particularly against fungi at low pH (0.1% w/v of sodium sulfite is used). Sodium sulfite is used in cosmetics, food products, and pharmaceutical applications such as parenteral formulations, inhalations, oral formulations, and topical preparations.
Sodium sulfite occurs as an odorless white powder or hexagonal prisms. Note that the commercially available sodium sulfite is often presented as a white to tan- or pink-colored powder that would not conform to the pharmacopeial specification.
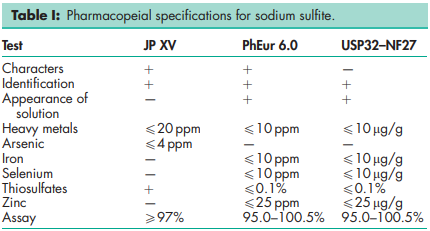
See Table I.
Acidity/alkalinity pH = 9 for an aqueous solution. Density 2.633 g/cm3 Hygroscopicity Hygroscopic. Solubility Soluble 1 in 3.2 parts of water; soluble in glycerin; practically insoluble in ethanol (95%).
Sodium sulfite should be stored in a well-closed container in a cool, dry, place. In solution, sodium sulfite is slowly oxidized to sulfate by dissolved oxygen; strong acids lead to formation of sulfurous acid/ sulfur dioxide. On heating, sodium sulfite decomposes liberating sulfur oxides
Sodium sulfite is incompatible with acids, oxidizing agents, many proteins, and vitamin B1.
Sodium bisulfite is prepared by reacting sulfur dioxide gas with sodium hydroxide solution. The solid material is obtained by evaporation of water. Further neutralization with sodium hydroxide while keeping the temperature above 33.68C leads to crystallization of the anhydrous sodium sulfite (below this temperature the heptahydrate form is obtained).
Sodium sulfite is widely used in food and pharmaceutical applications as an antioxidant. It is generally regarded as relatively nontoxic and nonirritant when used as an excipient.(2,3) However, contact dermatitis and hypersensitivity reactions have been reported.(4,5) The acceptable daily intake for sodium sulfite has been set at up to 350 mg/kg body-weight daily.(6) LD50 (mouse, IP): 0.950 g/kg(7) LD50 (mouse, IV): 0.130 g/kg LD50 (mouse, oral): 0.820 g/kg LD50 (rabbit, IV): 0.065 g/kg LD50 (rabbit, oral): 1.181 g/kg LD50 (rat, IV): 0.115 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled.
GRAS listed. Accepted for use as a food additive in Europe. Included in FDA Inactive Ingredients Database (epidural, IM, IV, and SC injections; inhalation solution; ophthalmic solutions; oral syrups and suspensions; otic solutions; topical creams and emulsions). Included in nonparenteral medicines licensed in the UK
Sodium sulfite heptahydrate; sodium metabisulfite Sodium sulfite heptahydrate Synonyms Natrii sulfis heptahydricus CAS number [7785-83-7] Molecular weight 252.15 Description Colorless crystals. Density 1.56 g/cm3 Solubility 1 in 1.6 of water; 1 in 30 of glycerin; sparingly soluble in ethanol (95%). Comments Sodium sulfite heptahydrate is included in the PhEur 6.0. The heptahydrate is unstable, oxidizing in the air to the sulfate.
A specification for sodium sulfite is contained in the Food Chemicals Codex (FCC).(8) The EINECS number for sodium sulfite is 231-821-4. The PubChem Compound ID (CID) for sodium sulfite is 24437.