

Name: Sorbic Acid
CAS No: (E,E)-Hexa-2,4-dienoic acid [22500-92-1]
BP: Sorbic Acid PhEur: Sorbic Acid USP-NF: Sorbic Acid
Acidum sorbicum; E200; (2-butenylidene) acetic acid; crotylidene acetic acid; hexadienic acid; hexadienoic acid; 2,4-hexadienoic acid; 1,3-pentadiene-1-carboxylic acid; 2-propenylacrylic acid; (E,E)- sorbic acid; Sorbistat K.
(E,E)-Hexa-2,4-dienoic acid [22500-92-1]
C6H8O2 112.13
Sorbic acid is an antimicrobial preservative(1) with antibacterial and antifungal properties used in pharmaceuticals, foods, enteral preparations, and cosmetics. Generally, it is used at concentrations of 0.05–0.2% in oral and topical pharmaceutical formulations, especially those containing nonionic surfactants. Sorbic acid is also used with proteins, enzymes, gelatin, and vegetable gums.(2) It has been shown to be an effective preservative for promethazine hydrochloride solutions in a concentration of 1 g/L.(3) Sorbic acid has limited stability and activity against bacteria and is thus frequently used in combination with other antimicrobial preservatives or glycols, when synergistic effects appear to occur;
Sorbic acid is a tasteless, white to yellow-white crystalline powder with a faint characteristic odor.
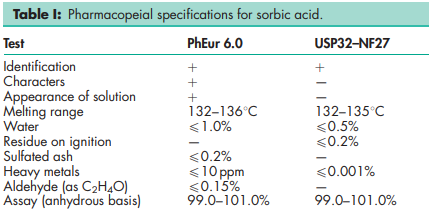
See Table I

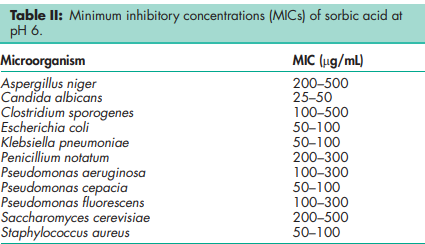
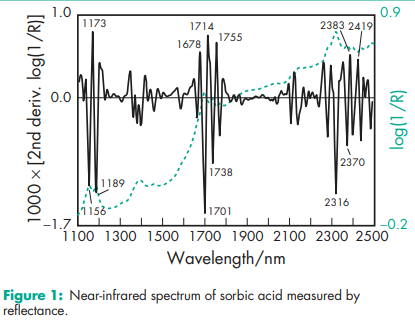
Antimicrobial activity Sorbic acid is primarily used as an antifungal agent, although it also possesses antibacterial properties. The optimum antibacterial activity is obtained at pH 4.5; and practically no activity is observed above pH 6.(4,5) The efficacy of sorbic acid is enhanced when it is used in combination with other antimicrobial preservatives or glycols since synergistic effects occur.(6) Reported minimum inhibitory concentrations (MICs) at pH 6 are shown in Table II.(7) Boiling point 2288C with decomposition. Density 1.20 g/cm3 Dissociation constant pKa = 4.76 Flash point 1278C Melting point 134.58C NIR spectra see Figure 1. Solubility see Table III. In syrup, the solubility of sorbic acid decreases with increasing sugar content. Vapor pressure <1.3 Pa (<0.01 mmHg) at 208C

Sorbic acid is sensitive to oxidation, particularly in the presence of light; oxidation occurs more readily in aqueous solution than in the solid form. Sorbic acid may be stabilized by phenolic antioxidants such as 0.02% propyl gallate.(6) Sorbic acid is combustible when exposed to heat or flame. When heated to decomposition, it emits acrid smoke and irritating fumes. The bulk material should be stored in a well-closed container, protected from light, at a temperature not exceeding 408C.
Sorbic acid is incompatible with bases, oxidizing agents, and reducing agents. Some loss of antimicrobial activity occurs in the presence of nonionic surfactants and plastics. Oxidation is catalyzed by heavy-metal salts. Sorbic acid will also react with sulfur-containing amino acids, although this can be prevented by the addition of ascorbic acid, propyl gallate, or butylhydroxytoluene. When stored in glass containers, the solution becomes very pH sensitive; therefore, preparations using sorbic acid as a preservative should be tested for their microbial purity after prolonged periods of storage. Aqueous solutions of sorbic acid without the addition of antioxidants are rapidly decomposed when stored in polypropylene, polyvinylchloride, and polyethylene containers.
Naturally occurring sorbic acid may be extracted as the lactone (parasorbic acid) from the berries of the mountain ash Sorbus aucuparia L. (Fam. Rosaceae). Synthetically, sorbic acid may be prepared by the condensation of crotonaldehyde and ketene in the presence of boron trifluoride; by the condensation of crotonaldehyde and malonic acid in pyridine solution; or from 1,1,3,5- tetraalkoxyhexane. Fermentation of sorbaldehyde or sorbitol with bacteria in a culture medium has also been used.
Sorbic acid is used as an antimicrobial preservative in oral and topical pharmaceutical formulations and is generally regarded as a nontoxic material. However, adverse reactions to sorbic acid and potassium sorbate, including irritant skin reactions(8–11) and allergic hypersensitivity skin reactions (which are less frequent), have been reported.(12–14) Other adverse reactions that have been reported include exfoliative dermatitis due to ointments that contain sorbic acid,(15) and allergic conjunctivitis caused by contact lens solutions preserved with sorbic acid.(16) No adverse reactions have been described after systemic administration of sorbic acid, and it has been reported that it can be ingested safely by patients who are allergic to sorbic acid.(17) However, perioral contact urticaria has been reported.(11) The WHO has set an estimated total acceptable daily intake for sorbic acid, calcium sorbate, potassium sorbate, and sodium sorbate, expressed as sorbic acid, at up to 25 mg/kg bodyweight.(18,19) Animal toxicological studies have shown no mammalian carcinogenicity or teratogenicity for sorbic acid consumed at up to 10% of the diet.(20) LD50 (mouse, IP): 2.82 g/kg(21) LD50 (mouse, oral): 3.20 g/kg LD50 (mouse, SC): 2.82 g/kg LD50 (rat, oral): 7.36 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Sorbic acid can be irritant to the skin, eyes, and respiratory system. Eye protection, gloves, and a dust mask or respirator are recommended.
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (ophthalmic solutions; oral capsules, solutions, syrups, tablets; topical and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Calcium sorbate; potassium sorbate; sodium sorbate. Calcium sorbate Empirical formula C12H14O4Ca Synonyms E203 Molecular weight 262.33 CAS number [7492-55-9] Appearance White, odorless, tasteless, crystalline powder. Solubility Soluble 1 in 83 parts of water; practically insoluble in fats. Comments The EINECS number for calcium sorbate is 231-321- 6. Sodium sorbate Empirical formula C6H7O2Na Synonyms E201; sodium (E,E)-hexa-2,4-dienoate. Molecular weight 134.12 CAS number [42788-83-0] Appearance Light, white, crystalline powder. Solubility Soluble 1 in 3 parts of water. Comments The EINECS number for sodium sorbate is 231-819- 3.
The trans,trans-isomer of sorbic acid is the commercial product. A specification for sorbic acid is contained in the Food Chemicals Codex (FCC).(22) The EINECS number for sorbic acid is 203-768-7. The PubChem Compound ID (CID) for sorbic acid includes 643460 and 1550734.