

Name: Phenol
CAS No: Phenol [108-95-2]
BP: Phenol JP: Phenol PhEur: Phenol USP: Phenol
Carbolic acid; hydroxybenzene; oxybenzene; phenic acid; phenolum; phenyl hydrate; phenyl hydroxide; phenylic acid; phenylic alcohol.
Phenol [108-95-2]
C6H6O 94.11
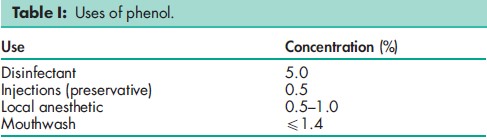
Phenol is used mainly as an antimicrobial preservative in parenteral pharmaceutical products. It has also been used in topical pharmaceutical formulations and cosmetics; see Table I. Phenol is widely used as an antiseptic, disinfectant, and therapeutic agent, although it should not be used to preserve preparations that are to be freeze-dried.(1)
Phenol occurs as colorless to light pink, caustic, deliquescent needleshaped crystals or crystalline masses with a characteristic odor. When heated gently phenol melts to form a highly refractive liquid. The USP 32 permits the addition of a suitable stabilizer; the name and amount of substance used for this purpose must be clearly stated on the label.
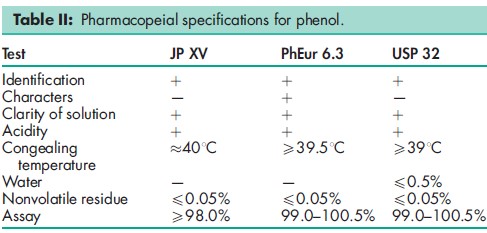
See Table II.
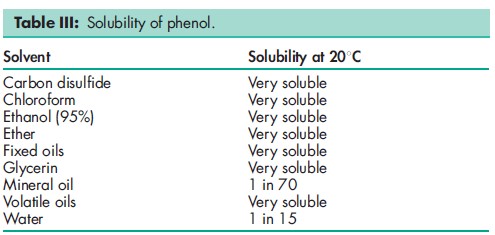
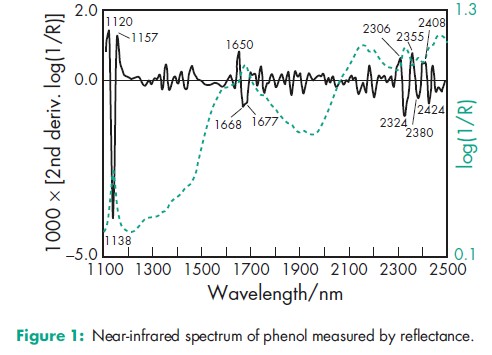
Acidity/alkalinity pH = 6.0 (saturated aqueous solution) Antimicrobial activity Phenol exhibits antimicrobial activity against a wide range of microorganisms such as Gram-negative and Gram-positive bacteria, mycobacteria and some fungi, and viruses; it is only very slowly effective against spores. Aqueous solutions of 1% w/v concentration are bacteriostatic, while stronger solutions are bactericidal. Phenol shows most activity in acidic solutions; increasing temperature also increases the antimicrobial activity. Phenol is inactivated by the presence of organic matter. Autoignition temperature 7158C Boiling point 181.88C Density 1.071 g/cm3 Dissociation constant pKa = 10 at 258C Flash point 798C (closed cup) Explosive limits 2% lower limit; 9% upper limit. Freezing point 40.98C Melting point 438C NIR spectra see Figure 1. Osmolarity A 2.8% w/v solution is iso-osmotic with serum. Partition coefficient Octanol : water = 1.46 Refractive index nD 41 = 1.5425 Solubility see Table III. Vapor density (relative) 3.24 (air = 1) Vapor pressure 133 Pa (1 mmHg) at 408C
When exposed to air and light, phenol turns a red or brown color, the color being influenced by the presence of metallic impurities. Oxidizing agents also hasten the color change. Aqueous solutions of phenol are stable. Oily solutions for injection may be sterilized in hermetically sealed containers by dry heat. The bulk material should be stored in a well-closed, light-resistant container at a temperature not exceeding 158C.
Phenol undergoes a number of chemical reactions characteristic of alcohols; however, it possesses a tautomeric enol structure that is weakly acidic. It will form salts with sodium hydroxide or potassium hydroxide, but not with their carbonates or bicarbonates. Phenol is a reducing agent and is capable of reacting with ferric salts in neutral to acidic solutions to form a greenish-colored complex. Phenol decolorizes dilute iodine solutions, forming hydrogen iodide and iodophenol; stronger solutions of iodine react with phenol to form the insoluble 2,4,6-triiodophenol. Phenol is incompatible with albumin and gelatin as they are precipitated. It forms a liquid or soft mass when triturated with compounds such as camphor, menthol, thymol, acetaminophen, phenacetin, chloral hydrate, phenazone, ethyl aminobenzoate, methenamine, phenyl salicylate, resorcinol, terpin hydrate, sodium phosphate, or other eutectic formers. Phenol also softens cocoa butter in suppository mixtures.
Historically, phenol was produced by the distillation of coal tar. Today, phenol is prepared by one of several synthetic methods, such as the fusion of sodium benzenesulfonate with sodium hydroxide followed by acidification; the hydrolysis of chlorobenzene by dilute sodium hydroxide at high temperature and pressure to give sodium phenate, which on acidification liberates phenol (Dow process); or the catalytic vapor-phase reaction of steam and chlorobenzene at 5008C (Raschig process).
Phenol is highly corrosive and toxic, the main effects being on the central nervous system. The lethal human oral dose is estimated to be 1 g for an adult. Phenol is absorbed from the gastrointestinal tract, skin, and mucous membranes, and is metabolized to phenylglucuronide and phenyl sulfate, which are excreted in the urine. Although there are a number of reports describing the toxic effects of phenol, these largely concern instances of accidental poisoning(2,3) or adverse reactions during its use as a therapeutic agent.(4,5) Adverse reactions associated with phenol used as a preservative are less likely owing to the smaller quantities that are used; however, it has been suggested that the body burden of phenol should not exceed 50 mg in a 10-hour period.(6) This amount could be exceeded following administration of large volumes of phenolpreserved medicines. LD50 (mouse, IV): 0.11 g/kg(7) LD50 (mouse, oral): 0.3 g/kg LD50 (rabbit, skin): 0.85 g/kg LD50 (rat, skin): 0.67 g/kg LD50 (rat, oral): 0.32 g/kg LD50 (rat, SC): 0.46 g/kg
Phenol is toxic on contact with the skin or if swallowed or inhaled. Phenol is strongly corrosive, producing possibly irreversible damage to the cornea and severe skin burns, although the skin burns are painless owing to the anesthetic effects of phenol. Phenol should be handled with caution, particularly when hot, owing to the release of corrosive and toxic fumes. The use of fume cupboards, enclosed plants, or other environmental containment is recommended. Protective polyvinyl chloride or rubber clothing is recommended, together with gloves, eye protection, and respirators. Spillages on the skin or eyes should be washed with copious amounts of water. Affected areas of the skin should be washed with water followed by application of a vegetable oil. Medical attention should be sought. Phenol poses a slight fire hazard when cold and a moderate hazard when hot and exposed to heat or flame. In the UK, the workplace exposure limits for phenol are 2 ppm long-term (8-hour TWA).(8) In the USA, the permissible exposure limit is 19 mg/m3 long-term and the recommended exposure limits are 20 mg/m3 long-term, and a maximum of 60 mg/m3 short-term.
Included in the FDA Inactive Ingredients Database (injections). Included in medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Liquefied phenol. Liquefied phenol Appearance Liquefied phenol is phenol maintained as a liquid by the presence of approximately 10% water. It is a colorless liquid, with a characteristic aromatic odor, which may develop a red coloration on exposure to air and light. Specific gravity 1.065 at 258C Comments Liquefied phenol is often more convenient to use in a formulation than the crystalline form. However, liquefied phenol should not be used with fixed or mineral oils, although the crystalline solid may be used. Caution should be observed when handling liquified phenol to avoid contact with skin, as this could cause serious burns.
Although phenol is soluble in approximately 12 parts of water at ambient temperatures, larger amounts of phenol in water produce a two-phase system of phenol solution floating on a lower layer of wet phenol. At 208C, 100 parts of phenol may be liquefied by the addition of 10 parts of water. At 848C phenol is miscible with water in all proportions. The EINECS number for phenol is 203-632-7. The PubChem Compound ID (CID) for phenol is 996.