

Name: Soybean Oil
CAS No: Soybean oil [8001-22-7]
BP: Refined Soya Oil JP: Soybean Oil PhEur: Soya-Bean Oil, Refined USP: Soybean Oil
Aceite de soja; Calchem IVO-114; Lipex 107; Lipex 200; Shogun CT; soiae oleum raffinatum; soja bean oil; soyabean oil; soya bean oil.
Soybean oil [8001-22-7]
A typical analysis of refined soybean oil indicates the composition of the acids, present as glycerides, to be: linoleic acid 50–57%; linolenic acid 5–10%; oleic acid 17–26%; palmitic acid 9–13%; and stearic acid 3–6%. Other acids are present in trace quantities.(1)
See Sections 4 and 8.
In pharmaceutical preparations, soybean oil emulsions are primarily used as a fat source in total parenteral nutrition (TPN) regimens.(2) Although other oils, such as peanut oil, have been used for this purpose, soybean oil is now preferred because it is associated with fewer adverse reactions. Emulsions containing soybean oil have also been used as vehicles for the oral and intravenous administration of drugs;(3,4) drug substances that have been incorporated into such emulsions include amphotericin,(5–7) diazepam, retinoids,(8) vitamins,(9) poorly water-soluble steroids,(10,11) fluorocarbons,(12,13) ibuprofen,(14) and insulin.(15) In addition, soybean oil has been used in the formulation of many drug delivery systems such as liposomes,(16) microspheres,(17) dry emulsions,(18) self-emulsifying systems,(19,20) microemulsions,(21,22) nanoemulsions(23,24) and nanocapsules,(23) solid-in-oil suspensions,(25) and multiple emulsions.(26) Soybean oil may also be used in cosmetics and is consumed as an edible oil. As soybean oil has emollient properties, it is used as a bath additive in the treatment of dry skin conditions.
The USP 32 describes soybean oil as the refined fixed oil obtained from the seeds of the soya plant Glycine max Merr. (Fabaceae); if an antoxidant is added, the name and quantity must be specified on the label. The PhEur 6.2 defines refined soybean oil as the fatty oil obtained from the seeds of Glycine soja Sieb. and Zucc. and Glycine max (L.) Merr. (G. hispida (Moench) Maxim.) by extraction and subsequent refining; it may contain a suitable antioxidant. The PhEur 6.2 also includes a monograph for hydrogenated soybean oil. See Vegetable Oil, hydrogenated, type 1. Soybean oil is a clear, pale-yellow colored, odorless or almost odorless liquid, with a bland taste that solidifies between 10 and 168C.
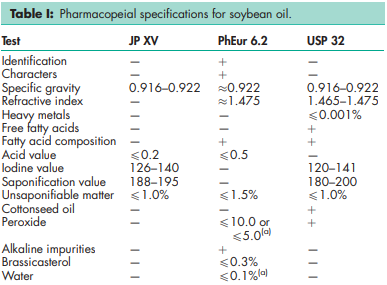
See Table I
Autoignition temperature 4458C Density 0.916–0.922 g/cm3 at 258C Flash point 2828C Freezing point 10 to 168C Hydroxyl value 4–8 Interfacial tension 50 mN/m (50 dynes/cm) at 208C. Refractive index nD 25 = 1.471–1.475 Solubility Practically insoluble in ethanol (95%) and water; miscible with carbon disulfide, chloroform, ether, and light petroleum. Surface tension 25 mN/m (25 dynes/cm) at 208C. Viscosity (dynamic) 172.9 mPa s (172.9 cP) at 08C; 99.7 mPa s (99.7 cP) at 108C; 50.09 mPa s (50.09 cP) at 258C; 28.86 mPa s (28.86 cP) at 408C.
Soybean oil is a stable material if protected from atmospheric oxygen. The formation of undesirable flavors in soybean oil is accelerated by the presence of 0.01 ppm copper and 0.1 ppm iron, which act as catalysts for oxidation; this can be minimized by the addition of chelating agents. Prolonged storage of soybean oil emulsions, particularly at elevated temperatures, can result in the formation of free fatty acids, with a consequent reduction in the pH of the emulsion; degradation is minimized at pH 6–7. However, soybean oil emulsions are stable at room temperature if stored under nitrogen in a light-resistant glass container. Plastic containers are permeable to oxygen and should not be used for long-term storage since oxidative degradation can occur. The stability of soybean oil emulsions is considerably influenced by other additives in a formulation.(27–33) Soybean oil should be stored in a well-filled, airtight, lightresistant container at a temperature not exceeding 258C.
Soybean oil emulsions have been reported to be incompatible at 258C with a number of materials including calcium chloride, calcium gluconate, magnesium chloride, phenytoin sodium, and tetracycline hydrochloride.(34) Lower concentrations of these materials, or lower storage temperatures, may result in improved compatibility. The source of the material may also affect compatibility; for example, while one injection from a particular manufacturer might be incompatible with a fat emulsion, an injection with the same amount of active drug substance from another manufacturer might be compatible. Amphotericin B has been reported to be incompatible with soybean oil containing fat emulsions under certain conditions.(35) Soybean oil emulsions are also incompatible with many other drug substances, IV infusion solutions, and ions (above certain concentrations). When plastic syringes are used to store soybean oil emulsion, silicone oil may be extracted into the emulsion; swelling of the syringe pump also occurs, resulting in the necessity for increased forces to maintain the motion of the plunger.(36
Obtained by solvent extraction using petroleum hydrocarbons, or to a lesser extent by expression using continuous screw-press operations, of the seeds of either Glycine max (Leguminosae) or Glycine soja (Leguminosae). The oil is refined, deodorized, and clarified by filtration at about 08C. Any phospholipids or sterols present are removed by refining with alkali.
Soybean oil is widely used intramuscularly as a drug vehicle or as a component of emulsions used in parenteral nutrition regimens; it is also consumed as an edible oil. Generally, soybean oil is regarded as an essentially nontoxic and nonirritant material. However, serious adverse reactions to soybean oil emulsions administered parenterally have been reported. These include cases of hypersensitivity,(37) CNS reactions,(38) and fat embolism.(39) Interference with the anticoagulant effect of warfarin has also been reported.(40) Anaphylactic reactions have also been reported following the consumption of foods derived from, or containing, soybeans. Recently there has been concern at the concentration of phytoestrogens in some soy-derived products. Administration of soy protein to humans has resulted in significantly decreased serum lipid concentrations.(41) In 1999, the UK Medical Devices Agency announced the voluntary withdrawal of a breast implant that contained soybean oil. The decision was taken because not enough was known at that time about the long-term safety and the rate of breakdown of the soybean oil in the filling and its possible effects on the body.(42) LD50 (mouse, IV): 22.1 g/kg(43) LD50 (rat, IV): 16.5 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Spillages of soybean oil are slippery and should be covered with an inert absorbent material prior to disposal.
Included in the FDA Inactive Ingredients Database (IV injections, oral capsules, and topical preparations). Included in nonparenteral (chewable tablets; oral capsules; oral lozenges; topical bath additives) and parenteral (emulsions for IV injection or infusion) medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Canola oil; corn oil; cottonseed oil; peanut oil; sesame oil; sunflower oil.
The stability of soybean oil emulsions may be readily disturbed by the addition of other materials, and formulations containing soybean oil should therefore be evaluated carefully for their compatibility and stability. A specification for soybean oil is contained in the Food Chemicals Codex (FCC).(44)