

Name: Starch, Pregelatinized
CAS No: Pregelatinized starch [9005-25-8]
BP: Pregelatinised Starch PhEur: Starch, Pregelatinised USP-NF: Pregelatinized Starch
Amylum pregelificatum; compressible starch; C*PharmGel; Instastarch; Lycatab C; Lycatab PGS; Merigel; National 78-1551; Pharma-Gel; Prejel; Sepistab ST200; Spress B820; Starch 1500 G; Tablitz; Unipure LD; Unipure WG220.
Pregelatinized starch [9005-25-8]
(C6H10O5)n where n = 300–1000. Pregelatinized starch is a starch that has been chemically and/or mechanically processed to rupture all or part of the starch granules. Both fully and partially pregelatinized grades are commercially available. Partial pregelatinization renders the starch flowable and directly compressible. Full pregelatinization produces a cold-water soluble starch that can be used as a wet granulation binder. Typically, pregelatinized starch contains 5% of free amylose, 15% of free amylopectin, and 80% unmodified starch. The USP32–NF27 does not specify the botanical origin of the original starch, but the PhEur 6.3 specifies that pregelatinized starch is obtained from maize (corn), potato, or rice starch. See also Starch and Section 13. Normally the fully pregelatinized starch contains 20–30% amylose and the rest amylopectin, which is about the same ratio (1 : 3) as for the partially pregelatinized form. There are ways to increase the amylose portion.(1)
See Starch.
Partially pregelatinized starch is a modified starch used in oral capsule and tablet formulations as a binder, diluent,(2,3) and disintegrant.(4) In comparison to starch, partially pregelatinized starch may be produced with enhanced flow and compression characteristics such that the pregelatinized material may be used as a tablet binder in dry-compression or direct compression processes.(5–15) In such processes, pregelatinized starch is self-lubricating. However, when it is used with other excipients it may be necessary to add a lubricant to a formulation. Although magnesium stearate 0.25% w/w is commonly used for this purpose, concentrations greater than this may have adverse effects on tablet strength and dissolution. Therefore, stearic acid is generally the preferred lubricant with pregelatinized starch.(16) Partially pregelatinized starch is used in oral dry powder hard capsule formulations. Both partially and fully pregelatinized starch may also be used in wet granulation processes.(17) See Table I. Fully pregelatinized starches can be used to make soft capsules, shells, and coatings as well as binders in tablets.

Pregelatinized starch occurs as a moderately coarse to fine, white to off-white colored powder. It is odorless and has a slight characteristic taste. Examination of fully pregelatinized starch as a slurry in cold water, under a polarizing microscope, reveals no significant ungelatinized granules, i.e. no ‘maltese crosses’ characteristic of the starch birefringence pattern. Examination of samples suspended in glycerin shows characteristic forms depending upon the method of drying used during manufacture: either irregular chunks from drum drying or thin plates. Partially pregelatinized starch (e.g. Starch 1500G and Sepistab ST200) show retention of birefringence patterns typical of unmodified starch granules.

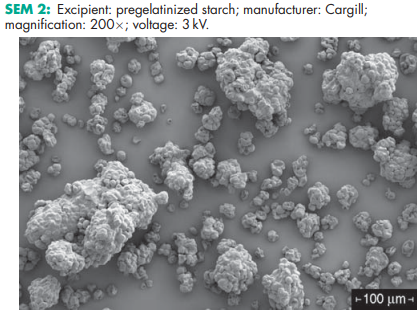


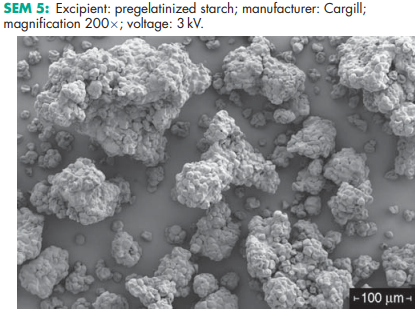
See Table II
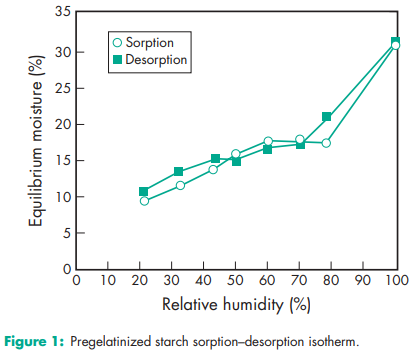
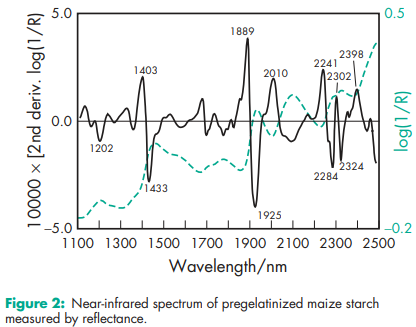
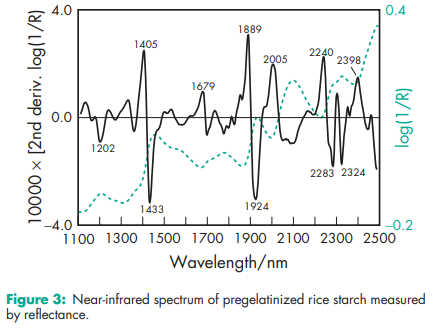
Acidity/alkalinity pH = 4.5–7.0 for a 10% w/v aqueous dispersion. Angle of repose 40.78 (6) Density (bulk) 0.586 g/cm Density (tapped) 0.879 g/cm3 Density (true) 1.516 g/cm3 Flowability 18–23% (Carr compressibility index)(18) Moisture content Pregelatinized maize starch is hygroscopic.(15,19,20) See also Figure 1. NIR spectra see Figures 2 and 3. Particle size distribution 30–150 mm, median diameter 52 mm. For partially pregelatinized starch, greater than 90% through a US #100 mesh (149 mm); and less than 0.5% retained on a US #40 mesh (420 mm). Solubility Practically insoluble in organic solvents. Slightly soluble to soluble in cold water, depending upon the degree of pregelatinization. Pastes can be prepared by sifting the pregelatinized starch into stirred, cold water. Cold-water-soluble matter for partially pregelatinized starch is 10–20%. Specific surface area 0.26 m2 /g (Colorcon); 0.18–0.28 m2 /g (Roquette). Viscosity (dynamic) 8–10 mPa s (8–10 cP) for a 2% w/v aqueous dispersion at 258C.
Pregelatinized starch is a stable but hygroscopic material, which should be stored in a well-closed container in a cool, dry place.
Food-grade pregelatinized starches are prepared by heating an aqueous slurry containing up to 42% w/w of starch at 62–728C. Chemical additives that may be included in the slurry are gelatinization aids (salts or bases) and surfactants, added to control rehydration or minimize stickiness during drying. After heating, the slurry may be spray-dried, roll-dried, extruded, or drum-dried. In the last case, the dried material may be processed to produce a desired particle size range. Pharmaceutical grades of fully pregelatinized starch use no additives and are prepared by spreading an aqueous suspension of ungelatinized starch on hot drums where gelatinization and subsequent drying take place. Partially pregelatinized starch is produced by subjecting moistened starch to mechanical pressure. The resultant material is ground and the moisture content is adjusted to specifications.
Pregelatinized starch and starch are widely used in oral solid-dosage formulations. Pregelatinized starch is generally regarded as a nontoxic and nonirritant excipient. However, oral consumption of large amounts of pregelatinized starch may be harmful. See Starch for further information.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and a dust mask are recommended. Excessive dust generation should be avoided to minimize the risks of explosions. In the UK, the long-term (8-hour TWA) workplace exposure limits for starch are 10 mg/m3 for total inhalable dust and 4 mg/m3 for respirable dust.(21
Included in the FDA Inactive Ingredients Database (oral capsules, suspensions, and tablets; vaginal preparations). Included in nonparenteral medicines licensed in the UK.
Corn starch and pregelatinized starch; starch; starch, sterilizable maize.
Pregelatinized starch is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. The USP32–NF27 also lists pregelatinized modified starch. A low-moisture grade of pregelatinized starch, Starch 1500 LM (Colorcon), containing less than 7% of water, specifically intended for use as a diluent in capsule formulations is commercially available.(16) Sepistab ST200 is described as an agglomerate of starch granules consisting of native and pregelatinized corn starch.(22) Compression characteristics of pregelatinized starches from sorghum and plantain have been evaluated against traditional corn-based products.(23) StarCap 1500 (Colorcon) is a coprocessed mixture of pregelatinized starch and corn starch promoted for use in dry-powder, hard-capsule fillings; see Corn Starch and Pregelatinized Starch