

Name: Starch, Sterilizable Maize
CAS No: Sterilizable maize starch
USP: Absorbable Dusting Powde
Bio-sorb; double-dressed, white maize starch; Fluidamid R444P; Keoflo ADP; Meritena; modified starch dusting powder; Pure-Dent B851; starch-derivative dusting powder; sterilizable corn starch.
Sterilizable maize starch
(C6H10O5)n where n = 300–1000.
See Starch
Sterilizable maize starch is a chemically or physically modified corn (maize) starch that does not gelatinize on exposure to moisture or steam sterilization. Sterilizable maize starch is primarily used as a lubricant for examination and surgeons’ gloves, although because of safety concerns unlubricated gloves are now generally recommended; see Section 14. It is also used as a vehicle for medicated dusting powders
Sterilizable maize starch occurs as an odorless, white, free-flowing powder. Particles may be rounded or polyhedral in shape
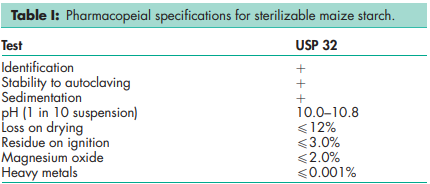
See Table I.
Acidity/alkalinity pH = 9.5–10.8 for a 10% w/v suspension at 258C. Density 1.48 g/cm3 Density (bulk) 0.47–0.59 g/cm Density (tapped) 0.64–0.83 g/cm3 Flowability 24–30% (Carr compressibility index)(1) Moisture content 10–15% Particle size distribution 6–25 mm; median diameter is 16 mm. Solubility Very slightly soluble in chloroform and ethanol (95%); practically insoluble in water. Specific surface area 0.50–1.15 m2 /g
Sterilizable maize starch may be sterilized by autoclaving at 1218C for 20 minutes, by ethylene oxide, or by irradiation.(2
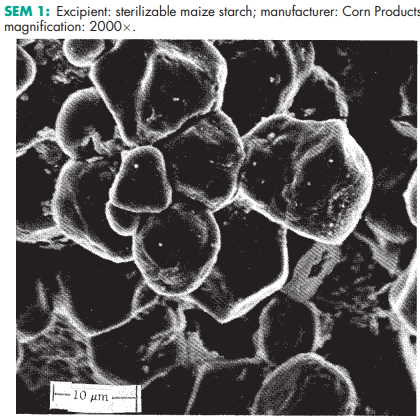
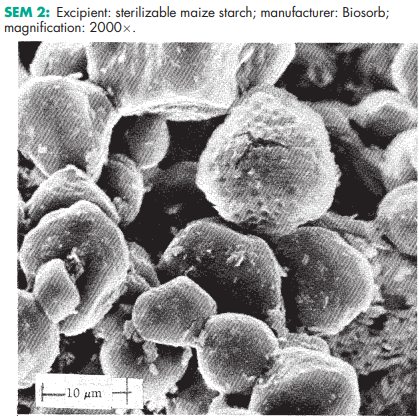

Corn starch (maize starch) is physically or chemically modified by treatment with either phosphorus oxychloride or epichlorhydrin so that the branched-chain and straight-chain starch polymers crosslink. Up to 2.0% of magnesium oxide may also be added to the starch.
Sterilizable maize starch is primarily used as a lubricant for surgeons’ gloves and as a vehicle for topically applied dusting powders. Granulomatous reactions, peritonitis and inflammation at operation sites have been attributed to contamination with surgical glove powders containing sterilizable maize starch. In addition, glove powder may be a risk factor in the development of latex allergy. As a consequence, it has been suggested that the use of sterilizable maize starch in latex gloves should be prohibited.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and a dust mask are recommended. Excessive dust generation should be avoided to minimize the risks of explosions. In the UK, the long-term (8-hour TWA) occupational exposure limits for starch are 10 mg/m3 for total inhalable dust and 4 mg/m3 for respirable dust.(13
Included in the FDA Inactive Ingredients Database (oral tablets and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Starch; starch, pregelatinized.