

Name: Phenylmercuric Nitrate
CAS No: There are two CAS Registry Numbers associated with phenylmercuric nitrate. One refers to the mixture of phenylmercuric nitrate and phenylmercuric hydroxide (C12H11Hg2NO4) while the other refers to phenylmercuric nitrate alone (C6H5HgNO3). The PhEur 6.0, and USP32–NF27 use the name phenylmercuric nitrate to describe the mixture and use the CAS Registry Number [55-68-5]. Hydroxyphenylmercury mixture with (nitrato-O)phenylmercury: C12H11Hg2NO4 [8003-05-2] (Nitrato-O)phenylmercury: C6H5HgNO3 [55-68-5]
BP: Phenylmercuric Nitrate PhEur: Phenylmercuric Nitrate USP-NF: Phenylmercuric Nitrate
Basic phenylmercury nitrate; mercuriphenyl nitrate; merphenyl nitrate; nitratophenylmercury; phenylhydrargyri nitras; phenylmercury nitrate; Phe-Mer-Nite; PMN. Note that the synonyms above are usually used to refer to phenylmercuric nitrate alone. However, confusion with nomenclature and CAS Registry Number has led to these synonyms also being applied to the PhEur 6.0 and USP32–NF27 material, which is a compound of phenylmercuric nitrate and phenylmercuric hydroxide.
There are two CAS Registry Numbers associated with phenylmercuric nitrate. One refers to the mixture of phenylmercuric nitrate and phenylmercuric hydroxide (C12H11Hg2NO4) while the other refers to phenylmercuric nitrate alone (C6H5HgNO3). The PhEur 6.0, and USP32–NF27 use the name phenylmercuric nitrate to describe the mixture and use the CAS Registry Number [55-68-5]. Hydroxyphenylmercury mixture with (nitrato-O)phenylmercury: C12H11Hg2NO4 [8003-05-2] (Nitrato-O)phenylmercury: C6H5HgNO3 [55-68-5]
C12H11Hg2NO4 634.45
Phenylmercuric salts are used as antimicrobial preservatives mainly in ophthalmic preparations, but are also used in cosmetics (see Section 16), parenteral, and topical pharmaceutical formulations; see Table I. Phenylmercuric salts are active over a wide pH range against bacteria and fungi and are usually used in neutral to alkaline solutions, although they have also been used effectively at slightly acid pH; see Section 10. In acidic formulations, phenylmercuric nitrate may be preferred to phenylmercuric acetate or phenylmercuric borate as it does not precipitate. Phenylmercuric nitrate is also an effective spermicide, although its use in vaginal contraceptives is no longer recommended; A number of adverse reactions to phenylmercuric salts have been reported, and concern at the toxicity of mercury compounds may preclude the use of phenylmercuric salts under certain circumstances;

Phenylmercuric nitrate PhEur 6.0, and USP32–NF27, is an equimolecular compound of phenylmercuric hydroxide and phenylmercuric nitrate; it occurs as a white, crystalline powder with a slight aromatic odor.
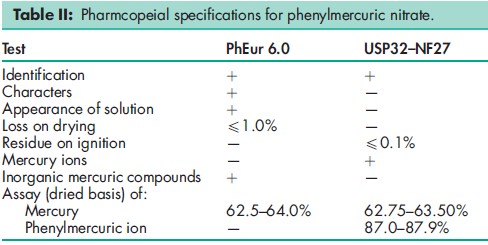
See Table II.
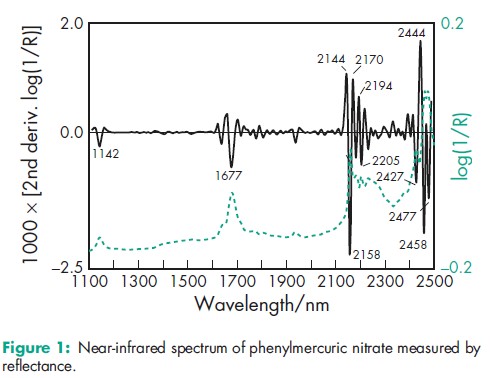
Acidity/alkalinity A saturated aqueous solution is acidic to litmus. Antimicrobial activity Phenylmercuric salts are broad-spectrum, growth-inhibiting agents at the concentrations normally used for the preservation of pharmaceuticals. They possess slow bactericidal and fungicidal activity. Antimicrobial activity tends to increase with increasing pH, although in solutions of pH 6 and below, activity against Pseudomonas aeruginosa has been demonstrated. Phenylmercuric salts are included in several compendial eye drop formulations of acid pH. Activity is also increased in the presence of phenylethyl alcohol, and in the presence of sodium metabisulfite at acid pH. Activity is decreased in the presence of sodium metabisulfite at alkaline pH.(1–3) When used as preservatives in topical creams, phenylmercuric salts are active at pH 5–8.(4) Bacteria (Gram-positive): Staphylococcus aureus Good inhibition, more moderate cidal activity. Minimum inhibitory concentration (MIC) against Staphylococcus aureus is 0.5 mg/mL. Bacteria (Gram-negative): Pseudomonas aeruginosa Inhibitory activity for most Gram-negative bacteria is similar to that for Gram-positive bacteria (MIC is approximately 0.3–0.5 mg/mL). Phenylmercuric salts are less active against some Pseudomonas species, and particularly Pseudomonas aeruginosa (MIC is approximately 12 mg/mL). Fungi: Candida albicans and Aspergillus niger Most fungi are inhibited by 0.3–1 mg/mL; phenylmercuric salts exhibit both inhibitory and fungicidal activity; e.g. for phenylmercuric acetate against Candida albicans, MIC is 0.8 mg/mL; for phenylmercuric acetate against Aspergillus niger, MIC is approximately 10 mg/mL. Spores Phenylmercuric salts may be active in conjunction with heat. The BP 1980 included heating at 1008C for 30 minutes in the presence of 0.002% w/v phenylmercuric acetate or phenylmercuric nitrate as a sterilization method. However, in practice this may not be sufficient to kill spores and heating with a bactericide no longer appears as a sterilization method in the BP 2009. Dissociation constant pKa = 3.3 Melting point 187–1908C with decomposition. NIR spectra see Figure 1. Partition coefficients Mineral oil : water = 0.58; Peanut oil : water = 0.4. Solubility More soluble in the presence of either nitric acid or alkali hydroxides. See Table III.



All phenylmercuric compound solutions form a black residue of metallic mercury when exposed to light or after prolonged storage. Solutions may be sterilized by autoclaving, although significant amounts of phenylmercuric salts may be lost, hence reducing preservative efficacy, owing to incompatibilities with packaging components or other excipients, e.g. sodium metabisulfite.(5–7) See Section 12. Phenylmercuric nitrate should be stored in a well-closed container, protected from light, in a cool, dry place.
The antimicrobial activity of phenylmercuric salts may be reduced in the presence of anionic emulsifying agents and suspending agents, tragacanth, starch, talc, sodium metabisulfite,(8) sodium thiosulfate,( 2) disodium edetate,(2) and silicates (bentonite, aluminum magnesium silicate, magnesium trisilicate, and kaolin).(9,10) Phenylmercuric salts are incompatible with halides, particularly bromides and iodides, as they form less-soluble halogen compounds. At concentrations of 0.002% w/v precipitation may not occur in the presence of chlorides. Phenylmercuric salts are also incompatible with aluminum and other metals, ammonia and ammonium salts, amino acids, and with some sulfur compounds, e.g. in rubber. Phenylmercuric salts are absorbed by rubber stoppers and some types of plastic packaging components; uptake is usually greatest to natural rubbers and polyethylene, and least to polypropylene.(11–16) Incompatibilities with some types of filter membranes may also result in loss of phenylmercuric salts following sterilization by filtration.(17)
Phenylmercuric nitrate is readily formed by heating benzene with mercuric acetate, and treating the resulting acetate with an alkali nitrate.(18)
Phenylmercuric nitrate and other phenylmercuric salts have been widely used as antimicrobial preservatives in parenteral and topical pharmaceutical formulations. However, concern over the use of phenylmercuric salts in pharmaceuticals has increased as a result of greater awareness of the toxicity of mercury and other mercury compounds. This concern must, however, be balanced by the effectiveness of these materials as antimicrobial preservatives and the low concentrations in which they are employed. Phenylmercuric salts are irritant to the skin at 0.1% w/w concentration in petrolatum.(19) In solution, they may give rise to erythema and blistering 6–12 hours after administration. In a modified repeated insult patch test, a 2% w/v solution was found to produce extreme sensitization of the skin.(20,21) Eye drops containing phenylmercuric nitrate as a preservative should not be used continuously for prolonged periods as mercurialentis, a brown pigmentation of the anterior capsule of the lens may occur. Incidence is 6% in patients using eye drops for greater than 6 years; however, the condition is not associated with visual impairment.(22,23) Cases of atypical band keratopathy have also been attributed to phenylmercuric nitrate preservative in eye drops.(24) Concern that the absorption of mercury from the vagina may be harmful has led to the recommendation that phenylmercuric nitrate should not be used in intravaginal formulations.(25) LD50 (mouse, IV): 27 mg/kg(26) LD50 (mouse, oral): 50 mg/kg LD50 (rat, SC): 63 mg/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Phenylmercuric nitrate may be irritant to the skin, eyes, and mucous membranes. Eye protection, gloves, and a respirator are recommended.
Included in the FDA Inactive Ingredients Database (parenteral and ophthalmic preparations). Included in parenteral products and eye drops in the EU. Included in the Canadian List of Acceptable Nonmedicinal Ingredients (ophthalmic, nasal and otic preparations only up to 0.002%; there must be no other suitable alternative preservative). Prohibited in first aid antiseptic drug products, antimicrobial diaper rash drug products and vaginal contraceptive drug products in the USA. Limited uses permitted in Japan and the EU for cosmetics (see Phenylmercuric Acetate).
Phenylmercuric acetate; phenylmercuric borate; thimerosal.
Phenylmercuric salts should be used in preference to benzalkonium chloride as a preservative for salicylates and nitrates and in solutions of salts of physostigmine and epinephrine that contain 0.1% sodium sulfite.