

Name: Acetone
CAS No: 2-Propanone [67-64-1]
BP: Acetone PhEur: Acetone USP-NF: Acetone
Acetonum; dimethylformaldehyde; dimethyl ketone; b-ketopropane; pyroacetic ether
2-Propanone [67-64-1]
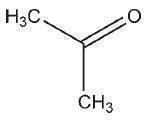
C3H6O 58.08
Acetone is used as a solvent or cosolvent in topical preparations, and as an aid in wet granulation.(1,2) It has also been used when formulating tablets with water-sensitive active ingredients, or to solvate poorly water-soluble binders in a wet granulation process. Acetone has also been used in the formulation of microspheres to enhance drug release.(3) Owing to its low boiling point, acetone has been used to extract thermolabile substances from crude drugs.(4)
Acetone is a colorless volatile, flammable, transparent liquid, with a sweetish odor and pungent sweetish taste.
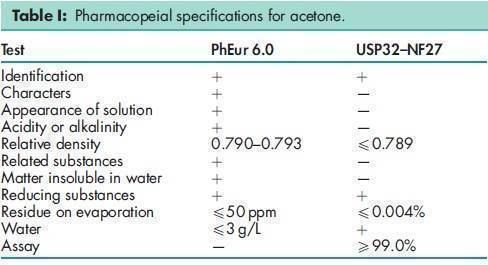
See Table I. See also Section 17.
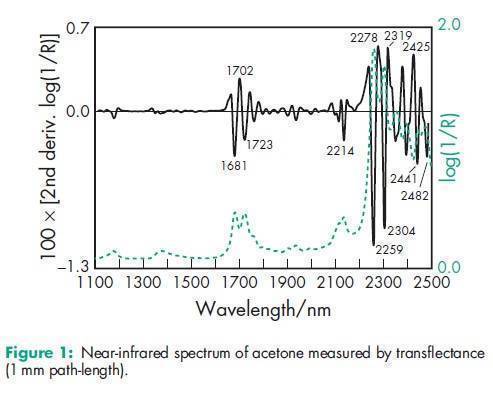
Boiling point 56.28C Flash point –208C Melting point 94.38C NIR spectra see Figure 1. Refractive index n D 20 = 1.359 Solubility Soluble in water; freely soluble in ethanol (95%). Vapor pressure 185mmHg at 208C
Acetone should be stored in a cool, dry, well-ventilated place out of direct sunlight.
Acetone reacts violently with oxidizing agents, chlorinated solvents, and alkali mixtures. It reacts vigorously with sulfur dichloride, potassium t-butoxide, and hexachloromelamine. Acetone should not be used as a solvent for iodine, as it forms a volatile compound that is extremely irritating to the eyes.(4)
Acetone is obtained by fermentation as a by-product of n-butyl alcohol manufacture, or by chemical synthesis from isopropyl alcohol; from cumene as a by-product in phenol manufacture; or from propane as a by-product of oxidation-cracking.
Acetone is considered moderately toxic, and is a skin irritant and severe eye irritant. Skin irritation has been reported due to its defatting action, and prolonged inhalation may result in headaches. Inhalation of acetone can produce systemic effects such as conjunctival irritation, respiratory system effects, nausea, and vomiting.(5) LD50 (mouse, oral): 3.0 g/kg(5) LD50 (mouse, IP): 1.297 g/kg LD50 (rabbit, oral): 5.340 g/kg LD50 (rabbit, skin): 0.2 g/kg LD50 (rat, IV): 5.5 g/kg LD50 (rat, oral): 5.8 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Acetone is a skin and eye irritant (see Section 14); therefore gloves, eye protection and a respirator are recommended. In the UK, the long-term (8-hour TWA) workplace exposure limit for acetone is 1210 mg/m3 (500 ppm). The shortterm (15-minute) exposure limit is 3620 mg/m3 (1500 ppm).(6)
Included in the FDA Inactive Ingredients Database (inhalation solution; oral tablets; topical preparations). Included in the Canadian List of Acceptable Non-medicinal Ingredients. Included in nonparenteral medicines licensed in the UK.
Not Found
A specification for acetone is included in the Japanese Pharmaceutical Excipients (JPE).(7) The EINECS number for acetone is 200-662-2. The PubChem Compound ID (CID) for acetone is 180.