

Name: Propylene Glycol
CAS No: 1,2-Propanediol [57-55-6], ()-1,2-Propanediol [4254-14-2] , (þ)-1,2-Propanediol [4254-15-3]
BP: Propylene Glycol JP: Propylene Glycol PhEur: Propylene Glycol USP: Propylene Glycol
1,2-Dihydroxypropane; E1520; 2-hydroxypropanol; methyl ethylene glycol; methyl glycol; propane-1,2-diol; propylenglycolum.
1,2-Propanediol [57-55-6], ()-1,2-Propanediol [4254-14-2] , (þ)-1,2-Propanediol [4254-15-3]
C3H8O2 , 76.09
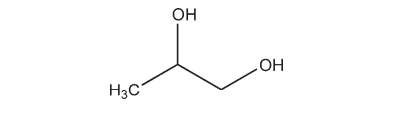
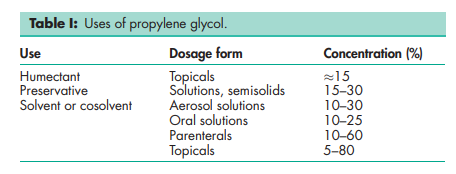
Propylene glycol has become widely used as a solvent, extractant, and preservative in a variety of parenteral and nonparenteral pharmaceutical formulations. It is a better general solvent than glycerin and dissolves a wide variety of materials, such as corticosteroids, phenols, sulfa drugs, barbiturates, vitamins (A and D), most alkaloids, and many local anesthetics. As an antiseptic it is similar to ethanol, and against molds it is similar to glycerin and only slightly less effective than ethanol. Propylene glycol is commonly used as a plasticizer in aqueous film-coating formulations. Propylene glycol is also used in cosmetics and in the food industry as a carrier for emulsifiers and as a vehicle for flavors in preference to ethanol, since its lack of volatility provides a more uniform flavor. See Table I.
Propylene glycol is a clear, colorless, viscous, practically odorless liquid, with a sweet, slightly acrid taste resembling that of glycerin.
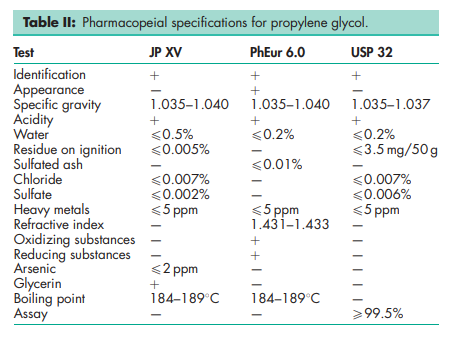
See Table II.
Autoignition temperature 3718C Boiling point 1888C Density 1.038 g/cm3 at 208C Flammability Upper limit, 12.6% v/v in air; lower limit, 2.6% v/v in air. Flash point 998C (open cup) Heat of combustion 1803.3 kJ/mol (431.0 kcal/mol) Heat of vaporization 705.4 J/g (168.6 cal/g) at b.p. Melting point 598C Osmolarity A 2.0% v/v aqueous solution is iso-osmotic with serum. Refractive index nD 20 = 1.4324 Specific rotation [a]D 20 = 15.08 (neat) for (R)-form; [a]D 20 = þ15.88 (neat) for (S)-form. Solubility Miscible with acetone, chloroform, ethanol (95%), glycerin, and water; soluble at 1 in 6 parts of ether; not miscible with light mineral oil or fixed oils, but will dissolve some essential oils. Specific heat 2.47 J/g (0.590 cal/g) at 208C. Surface tension 40.1 mN/m (40.1 dynes/cm) at 258C. Vapor density (relative) 2.62 (air = 1) Vapor pressure 9.33 Pa (0.07 mmHg) at 208C. Viscosity (dynamic) 58.1 mPa s (58.1 cP) at 208C.
At cool temperatures, propylene glycol is stable in a well-closed container, but at high temperatures, in the open, it tends to oxidize, giving rise to products such as propionaldehyde, lactic acid, pyruvic acid, and acetic acid. Propylene glycol is chemically stable when mixed with ethanol (95%), glycerin, or water; aqueous solutions may be sterilized by autoclaving. Propylene glycol is hygroscopic and should be stored in a wellclosed container, protected from light, in a cool, dry place.
Propylene glycol is incompatible with oxidizing reagents such as potassium permanganate.
Propylene is converted to chlorohydrin by chlorine water and hydrolyzed to 1,2-propylene oxide. With further hydrolysis, 1,2- propylene oxide is converted to propylene glycol.
Propylene glycol is used in a wide variety of pharmaceutical formulations and is generally regarded as a relatively nontoxic material. It is also used extensively in foods and cosmetics. Probably as a consequence of its metabolism and excretion, propylene glycol is less toxic than other glycols. Propylene glycol is rapidly absorbed from the gastrointestinal tract; there is also evidence that it is absorbed topically when applied to damaged skin. It is extensively metabolized in the liver, mainly to lactic and pyruvic acids, and is also excreted unchanged in the urine.(1,2) In topical preparations, propylene glycol is regarded as minimally irritant,(3) although it is more irritant than glycerin. There have been some reports of contact dermatitis associated with propylene glycol.(4,5) Some local irritation is produced upon application to mucous membranes or when it is used under occlusive conditions.(6) Parenteral administration may cause pain or irritation when propylene glycol is used in high concentration. Propylene glycol is estimated to be one-third as intoxicating as ethanol, with administration of large volumes being associated with adverse effects most commonly on the central nervous system, especially in neonates and children.(7–9) Other adverse reactions reported, though generally isolated, include: ototoxicity;(10) cardiovascular effects; seizures; and hyperosmolarity(11) and lactic acidosis, both of which occur most frequently in patients with renal impairment. Adverse effects are more likely to occur following consumption of large quantities of propylene glycol or on adminstration to neonates, children under 4 years of age, pregnant women, and patients with hepatic or renal failure. Adverse events may also occur in patients treated with disulfiram or metronidazole.(12) On the basis of metabolic and toxicological data, the WHO has set an acceptable daily intake of propylene glycol at up to 25 mg/kg body-weight.(13) Formulations containing 35% propylene glycol can cause hemolysis in humans. In animal studies, there has been no evidence that propylene glycol is teratogenic or mutagenic. Rats can tolerate a repeated oral daily dose of up to 30 mL/kg body-weight in the diet over 6 months, while the dog is unaffected by a repeated oral daily dose of 2 g/kg in the diet for 2 years.(14) LD50 (mouse, IP): 9.72 g/kg(15) LD50 (mouse, IV): 6.63 g/kg LD50 (mouse, oral): 22.0 g/kg LD50 (mouse, SC): 17.34 g/kg LD50 (rat, IM): 0.01 g/kg LD50 (rat, IP): 6.66 g/kg LD50 (rat, IV): 6.42 g/kg LD50 (rat, oral): 0.02 g/kg LD50 (rat, SC): 22.5 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Propylene glycol should be handled in a well-ventilated environment; eye protection is recommended. In the UK, the long-term (8-hour TWA) workplace exposure limit for propylene glycol vapor and particulates is 474 mg/m3 (150 ppm) and 10 mg/m3 for particulates.(16)
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (dental preparations; IM and IV injections; inhalations; ophthalmic, oral, otic, percutaneous, rectal, topical, and vaginal preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
Propylene glycol alginate.
Propylene glycol is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. In addition to its uses as an excipient, propylene glycol is used in veterinary medicine as an oral glucogenic in ruminants.(17) A specification for potassium glycol is contained in the Food Chemicals Codex (FCC).(18) The EINECS number for propylene glycol is 200-338-0. The PubChem Compound ID (CID) for propylene glycol is 1030.