

Name: Sucrose Octaacetate
CAS No: Acetic acid[(2S,3S,4R,5R), 4-acetoxy-2,5-bis(acetoxymethyl), 2- [[(2R,3R,4S,5R,6R). 3,4,5-triacetoxy-6-(acetoxymethyl), 2-tetrahydropyranyl]oxy], 3-tetrahydrofuranyl]ester [126-14-7]
USP-NF: Sucrose Octaacetate
a-D-Glucopyranoside, 1,3,4,6-tetra-O-acetyl-b-D-fructofuranosyl-, tetraacetate; octaacetylsucrose.
Acetic acid[(2S,3S,4R,5R), 4-acetoxy-2,5-bis(acetoxymethyl), 2- [[(2R,3R,4S,5R,6R). 3,4,5-triacetoxy-6-(acetoxymethyl), 2-tetrahydropyranyl]oxy], 3-tetrahydrofuranyl]ester [126-14-7]
C28H38O19 678.59
Sucrose octaacetate is used as an alcohol denaturant in pharmaceutical formulations. It is also used as a bittering agent, and is incorporated into preparations intended to deter nail-biting or thumb-sucking.
Sucrose octaacetate occurs as white hygroscopic powder. It is practically odorless with a bitter taste.
See Table I.

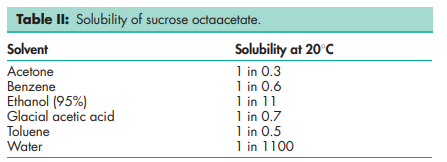
Boiling point 2608C Flash point 307.38C Melting point 898C (decomposes above 2858C) Refractive index 1.47 Solubility see Table II. Specific gravity 1.28 at 208C (water = 1) Specific rotation [a]D 25.4 = þ58.58
Sucrose octaacetate is a stable material and should be stored in a well-closed, airtight container. Store in a cool, dry place; moisture may cause instability.


Sucrose octaacetate is typically produced by chemical synthesis; one reported synthetic method is by pyridine-catalyzed acetylation of sucrose.(1,2)
Sucrose octaacetate is generally regarded as safe. It is considered slightly hazardous in cases of skin contact (irritant), ingestion, or inhalation.
Observe normal precautions appropriate to the circumstances and quantity of the material handled. When heated to decomposition, sucrose octaacetate emits acrid smoke and irritating vapors. Compatible chemical-resistant gloves and eye safety goggles are recommended. Respiratory protection is not required, but dust masks may be used for protection from nuisance levels of dust.
GRAS listed. Approved by the FDA as both a direct and an indirect food additive, and as a nail-biting deterrent for over-the-counter drug products.(4)
Sodium acetate; sucrose.
Sucrose octaacetate is a naturally occurring substance that has been isolated from plant material: the root of Clematis japonica contains 0.15% of sucrose octaacetate by dry weight. At a concentration of 0.06% sucrose octaacetate renders sugar too bitter for human consumption.(5) The EINECS number for sucrose octaacetate is 204-772-1. The PubChem Compound ID (CID) for sucrose octaacetate is 219904.