

Name: Sugar, Compressible
CAS No: See Sections 4 and 18.
BP: Compressible Sugar USP-NF: Compressible Sugar
Compressuc; Comprima; Di-Pac; direct compacting sucrose; directly compressible sucrose; Nu-Tab.
See Sections 4 and 18.
The BP 2009 and USP32–NF27 state that compressible sugar contains not less than 95.0% and not more than 98.0% of sucrose (C12H22O11). It may contain starch, maltodextrin, or invert sugar, and may contain a suitable lubricant.
See Section 4.
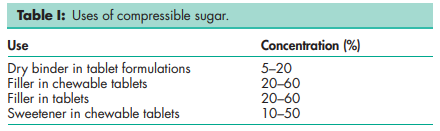
Compressible sugar is used primarily in the preparation of directcompression chewable tablets. Its tableting properties can be influenced by changes in moisture level;(1) see Table I for typical uses.
Compressible sugar is a free-flowing, sweet-tasting, white powder (or crystalline agglomerates).
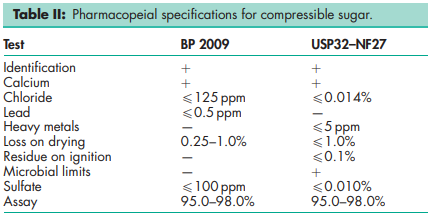
See Table II.

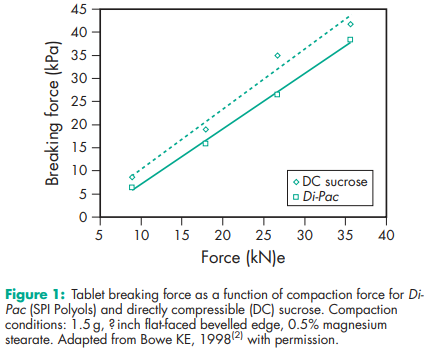
Compaction profile see Figure 1. Density (bulk) 0.609–0.673 g/cm3 for Di-Pac. Hygroscopicity Moisture content of Di-Pac depends on factors such as termperature, pressure, relative humidity, and time of exposure to a given environment.(3) Up to 80% relative humidity at 208C, water uptake is typically <1% w/w. Melting point 1868C for Di-Pac. Particle size distribution For Di-Pac, 3% maximum retained on a #40 (425 mm) mesh; 75% minimum through a #100 (150 mm) mesh; 5% maximum through #200 (75 mm) mesh. Solubility The sucrose portion is soluble in water. Specific surface area 0.13–0.14 m2 /g for Di-Pac.
Compressible sugar is physically stable at room temperature and low relative humidity. It deliquesces at above 80% relative humidity at 258C. The bulk material should be stored in a well-closed container in a cool, dry place.
Incompatible with dilute acids, which cause hydrolysis of sucrose to invert sugar, and with alkaline earth hydroxides, which react with sucrose to form sucrates.
Compressible sugar is prepared by cocrystallization of sucrose with other excipients such as maltodextrin.(1) Compressible sugar may also be prepared using a dry granulation process or fluid bed granulation process.
Compressible sugar is generally regarded as a relatively nontoxic and nonirritant material. See also Sucrose.
Observe normal precautions appropriate to the circumstances and quantity of material handled
Included in the FDA Inactive Ingredients Database (oral capsules and tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
Confectioner’s sugar; sucrose; sugar spheres; Sugartab. Sugartab Appearance Sugartab (JRS Pharma) is a compressible sugar that does not conform to the USP32–NF27 specification. It is an agglomerated sugar product containing approximately 90–93% sucrose, the balance being invert sugar. Density (bulk) 0.60 g/cm3 Density (tapped) 0.69 g/cm3 Flowability 42.7 g/s Moisture content 0.20–0.57%. Particle size distribution 30% through a #20 (850 mm) mesh; 3% through a #30 (600 mm) mesh.