

Name: Sugar Spheres
CAS No:
BP: Sugar Spheres PhEur: Sugar Spheres USP-NF: Sugar Spheres
Non-pareil; non-pareil seeds; Nu-Core; Nu-Pareil PG; sacchari sphaerae; sugar seeds; Suglets.
See Section 8.
See Section 8.
Sugar spheres are mainly used as inert cores in capsule and tablet formulations, particularly multiparticulate sustained-release formulations.(1–4) They form the base upon which a drug is coated, usually followed by a release-modifying polymer coating. Alternatively, a drug and matrix polymer may be coated onto the cores simultaneously. The active drug is released over an extended period either via diffusion through the polymer or through to the controlled erosion of the polymer coating. Complex drug mixtures contained within a single-dosage form may be prepared by coating the drugs onto different batches of sugar spheres with different protective polymer coatings. Sugar spheres are also used in confectionery products.
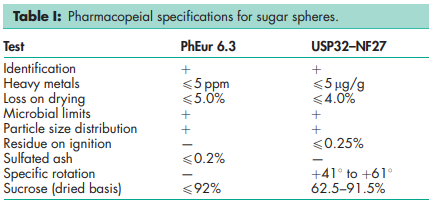
The USP32–NF27 describes sugar spheres as approximately spherical granules of a labeled nominal-size range with a uniform diameter and containing not less than 62.5% and not more than 91.5% of sucrose, calculated on the dried basis. The remainder is chiefly starch. The PhEur 6.3 states that sugar spheres contain not more than 92% of sucrose calculated on the dried basis. The remainder consists of corn (maize) starch and may also contain starch hydrolysates and color additives. The diameter of sugar spheres varies from 200 to 2000 mm, and the upper and lower limits of the size of the sugar spheres are stated on the label.
See Table I.
Density 1.57–1.59 g/cm3 for Suglets less than 500 mm in size; 1.55–1.58 g/cm3 for Suglets more than 500 mm in size. Flowability <10 seconds, free flowing. Particle size distribution Sugar spheres are of a uniform diameter. The following sizes are commercially available from various suppliers (US standard sieves): 45–60 mesh (250–355 mm) 40–50 mesh (300–425 mm) 35–45 mesh (355–500 mm) 35–40 mesh (420–500 mm) 30–35 mesh (500–600 mm) 25–30 mesh (610–710 mm) 20–25 mesh (710–850 mm) 18–20 mesh (850–1000 mm) 16–20 mesh (850–1180 mm) 14–18 mesh (1000–1400 mm) Solubility Solubility in water varies according to the sucrose-tostarch ratio. The sucrose component is freely soluble in water, whereas the starch component is practically insoluble in cold water. Specific surface area 0.1–0.2 m2 /g for Suglets less than 500 mm in size; >0.2 m2 /g for Suglets more than 500 mm in size.
Sugar spheres are stable when stored in a well-closed container in a cool, dry place.
See Starch and Sucrose for information concerning the incompatibilities of the component materials of sugar spheres.
Sugar spheres are prepared from crystalline sucrose, which is coated using sugar syrup and a starch dusting powder.
Sugar spheres are used in oral pharmaceutical formulations. The sucrose and starch components of sugar spheres are widely used in edible food products and oral pharmaceutical formulations. The adverse reactions and precautions necessary with the starch and sucrose components should be considered in any product containing sugar spheres. For example, sucrose is generall regarded as more cariogenic than other carbohydrates, and in higher doses is also contraindicated in diabetic patients.
Observe normal precautions appropriate to the circumstances and quantity of material handled.
Included in the FDA Inactive Ingredients Database (oral capsules and tablets). Included in nonparenteral medicines licensed in the UK and Europe. The sucrose and starch components of sugar spheres are individually approved for use as food additives in Europe and the USA. Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Compressible sugar; confectioner’s sugar; microcrystalline cellulose spheres; NPTAB; starch; sucrose. Microcrystalline cellulose spheres Comments Microcrystalline cellulose spheres are prepared from microcrystalline cellulose by spherization. They are available from several manufacturers and in different sizes, and the size grades can vary between the different manufacturers. Typical size grades available are: 120–230 mesh (63–125 mm); 70–140 mesh (106–212 mm); 30–100 mesh (150–300 mm); 45–70 mesh (212–355 mm); 35–45 mesh (355–500 mm); 25–35 mesh (500–710 mm); 18–25 mesh (710–1000 mm); 14–18 mesh (1000–1400 mm). NPTAB Appearance NPTAB (NP Pharm) is a compressible sugar that does not conform to the USP32–NF27 specification. It is an agglomerated sugar product containing not more than 92% sucrose, the balance being corn (maize) starch. Density 1.55–1.59 g/cm3 (varies with particle size) Flowability <10% (Carr compressibility index) Particle size distribution NPTAB 190 (180–212 mm); NPTAB 220 (212–250 mm); NPTAB 270 (250–300 mm); NPTAB 320 (300–350 mm).