

Name: Propyl Gallate
CAS No: 3,4,5-Trihydroxybenzoic acid propyl ester [121-79-9]
BP: Propyl Gallate PhEur: Propyl Gallate USP-NF: Propyl Gallate
E310; gallic acid propyl ester; n-propyl gallate; Progallin P; propyl 3,4,5-trihydroxybenzoate; propylis gallas; Tenox PG.
3,4,5-Trihydroxybenzoic acid propyl ester [121-79-9]
C10H12O5 , 212.20
Propyl gallate has become widely used as an antioxidant in cosmetics, perfumes, foods, and pharmaceuticals since its use in preventing autoxidation of oils was first described in 1943.(1,2) It is primarily used, in concentrations up to 0.1% w/v, to prevent the rancidity of oils and fats;(3) it may also be used at concentrations of 0.002% w/v to prevent peroxide formation in ether, and at 0.01% w/v to prevent the oxidation of paraldehyde. Synergistic effects with other antioxidants such as butylated hydroxyanisole and butylated hydroxytoluene have been reported. Propyl gallate is also said to possess some antimicrobial properties; see Section 10. Studies have shown that, when added to powder blends containing ketorolac, propyl gallate significantly increases the drug stability in the preparation.(4) Other alkyl gallates are also used as antioxidants and have approximately equivalent antioxidant properties when used in equimolar concentration; however, solubilities vary; see Section 17. Propyl gallate has also been investigated for its therapeutic properties, mainly in animal studies.
Propyl gallate is a white, odorless or almost odorless crystalline powder, with a bitter astringent taste that is not normally noticeable at the concentrations employed as an antioxidant.
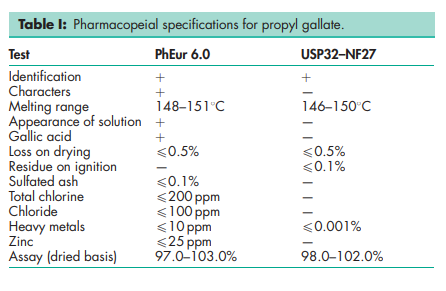
See Table I.
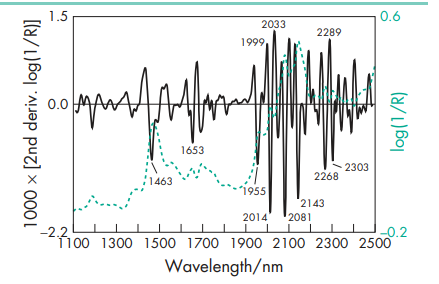
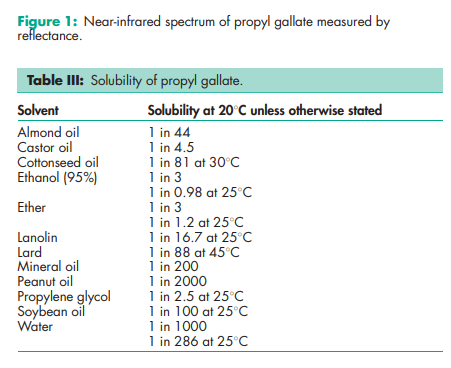
Acidity/alkalinity pH = 5.9 (0.1% w/v aqueous solution) Antimicrobial activity Propyl gallate has been reported to possess some antimicrobial activity against Gram-negative, Grampositive, and fungal species.(5) Its effectiveness as a preservative may be improved when used in combination with zinc salts, such as zinc sulfate, owing to synergistic effects.(6) For reported minimum inhibitory concentrations (MICs) for aqueous solutions containing 4% v/v ethanol as cosolvent, see Table II.(5) Dissociation constant pKa = 8.11 Melting point 1508C NIR spectra see Figure 1. Partition coefficients Octanol : water = 32; Oleyl alcohol : water = 17. Solubility see Table III.

Propyl gallate is unstable at high temperatures and is rapidly destroyed in oils that are used for frying purposes. The bulk material should be stored in a well-closed, nonmetallic container, protected from light, in a cool, dry place.
The alkyl gallates are incompatible with metals, e.g. sodium, potassium, and iron, forming intensely colored complexes. Complex formation may be prevented, under some circumstances, by the addition of a sequestering agent, typically citric acid. Propyl gallate may also react with oxidizing materials.
Propyl gallate is prepared by the esterification of 3,4,5-trihydroxybenzoic acid (gallic acid) with n-propanol. Other alkyl gallates are prepared similarl
It has been reported, following animal studies, that propyl gallate has a strong contact sensitization potential.(7) Propyl gallate has also produced cytogenic effects in CHO-K1 cells.(8) However, despite this, there have been few reports of adverse reactions to propyl gallate.(9) Those that have been described include contact dermatitis, allergic contact dermatitis,(9–13) and methemoglobinemia in neonates.(14) The WHO has set an estimated acceptable daily intake for propyl gallate at up to 1.4 mg/kg body-weight.(15) LD50 (cat, oral): 0.4 g/kg(16) LD50 (mouse, oral): 1.7 g/kg LD50 (rat, oral): 2.1 g/kg LD50 (rat, IP): 0.38 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended. When heated to decomposition, propyl gallate may emit toxic fumes and smoke.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IM injections; oral, and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
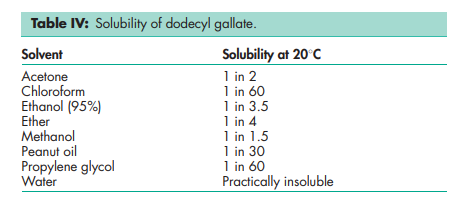
Dodecyl gallate; ethyl gallate; octyl gallate. Dodecyl gallate Empirical formula C19H30O5 Molecular weight 338.44 CAS number [1166-52-5] Synonyms Dodecyl 3,4,5-trihydroxybenzoate; dodecylis gallas; E312; lauryl gallate. Appearance White, odorless or almost odorless, crystalline powder. Melting point 96–97.58C Solubility see Table IV. Safety The WHO has established a temporary estimated acceptable daily intake for dodecyl gallate at up to 0.05 mg/kg bodyweight.(15) Comments The EINECS number for dodecyl gallate is 214-620-6. The PubChem Compound ID (CID) for dodecyl gallate is 14425. Ethyl gallate Empirical formula C9H10O5 Molecular weight 198.17 CAS number [831-61-8] Synonym Ethyl 3,4,5-trihydroxybenzoate Appearance White, odorless or almost odorless, crystalline powder. Melting point 151–1548C Solubility see Table V. Octyl gallate Empirical formula C15H22O5 Molecular weight 282.34 CAS number [1034-01-1] Synonyms E311; octyl 3,4,5-trihydroxybenzoate. Appearance White, odorless or almost odorless, crystalline powder. Melting point 100–1028C Solubility see Table VI. Safety The WHO has established a temporary estimated acceptable daily intake for octyl gallate at up to 0.1 mg/kg bodyweight.(15) Comments The EINECS number for octyl gallate is 252-073-5. The PubChem Compound ID (CID) for octyl gallate is 61253.

Propyl gallate has been reported to impart an ‘off’ flavor to corn and cottonseed oils when used as an antioxidant.(17) A specification for propyl gallate is contained in the Food Chemicals Codex (FCC).(18) The EINECS number for propyl gallate is 204-498-2