

Name: Phosphoric Acid
CAS No: Orthophosphoric acid [7664-38-2]
BP: Phosphoric Acid PhEur: Phosphoric Acid, Concentrated USP-NF: Phosphoric Acid
Acid fosforico; acide phosphorique; acidum phosphorum concentratum; E338; hydrogen phosphate; syrupy phosphoric acid.
Orthophosphoric acid [7664-38-2]
H3PO4 98.00
Phosphoric acid is widely used as an acidifying agent in a variety of pharmaceutical formulations. It is used in pharmaceutical products as part of a buffer system when combined with a phosphate salt such as sodium phosphate, monobasic or dibasic. It is also widely used in food preparations as an acidulant, flavor, and synergistic antioxidant (0.001–0.005%) and sequestrant. Therapeutically, dilute phosphoric acid has been used welldiluted in preparations used in the treatment of nausea and vomiting. Phosphoric acid 35% gel has also been used to etch tooth enamel and to enhance delivery of drugs through the nail.(3) Nanosized hydroxyapatite powder was made by combining phosphoric acid with egg shells.(4)
Concentrated phosphoric acid occurs as a colorless, odorless, syrupy liquid.
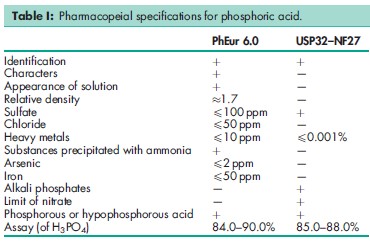
See Table I.
Acidity/alkalinity pH = 1.6 (1% w/w aqueous solution) Boiling point 117.878C Dissociation constant pKa1 = 2.15; pKa2 = 7.09; pKa3 = 12.32. Melting point 42.358C Refractive index nD 17.5 = 1.35846 (30% w/w aqueous solution); nD 17.5 = 1.35032 (20% w/w aqueous solution); nD 17.5 = 1.3423 (10% w/w aqueous solution). Solubility Miscible with ethanol (95%) and water with the evolution of heat. Specific gravity 1.874 (100% w/w) at 258C; 1.6850 (85% w/w aqueous solution) at 258C; 1.3334 (50% w/w aqueous solution) at 258C; 1.0523 (10% w/w aqueous solution) at 258C.
When stored at a low temperature, phosphoric acid may solidify, forming a mass of colorless crystals, comprising the hemihydrate, which melts at 288C. Phosphoric acid should be stored in an airtight container in a cool, dry place. Stainless steel containers may be used.
Phosphoric acid is a strong acid and reacts with alkaline substances. Mixtures with nitromethane are explosive.
The majority of phosphoric acid is made by digesting phosphate rock (essentially tricalcium phosphate) with sulfuric acid; the phosphoric acid is then separated by slurry filtration. Purification is achieved via chemical precipitation, solvent extraction, crystallization, or ion exchange.
In the concentrated form, phosphoric acid is an extremely corrosive and harmful acid. However, when used in pharmaceutical formulations it is usually very diluted and is generally regarded as an essentially nontoxic and nonirritant material. The lowest lethal oral dose of concentrated phosphoric acid in humans is reported to be 1286 mL/kg.(1) LD50 (rabbit, skin): 2.74 g/kg(1) LD50 (rat, oral): 1.53 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Phosphoric acid is corrosive and can cause burns on contact with the skin, eyes and mucous membranes; contact should be avoided. Splashes should be washed with copious quantities of water. Protective clothing, gloves and eye protection are recommended. Phosphoric acid is also irritant on inhalation. In the UK, the workplace exposure limit for phosphoric acid is 1 mg/m3 long-term (8-hour TWA) and 2 mg/m3 short-term (15-minutes).(2) Phosphoric acid emits toxic fumes on heating.
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (infusions, injections, oral solutions, topical creams, lotions, ointments and solutions, and vaginal preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Dilute phosphoric acid. Dilute phosphoric acid Synonyms acidum phosphoricum dilutum; diluted phosphoric acid. Comments The PhEur 6.0 states that dilute phosphoric acid contains 9.5–10.5% w/w H3PO4 and may be prepared by mixing phosphoric acid 115 g with 885 g of water. The USP32– NF27 contains a monograph for diluted phosphoric acid and states that it contains 9.5–10.5% w/v H3PO4 and may be prepared by mixing phosphoric acid 69mL with water to 1000 mL.
In the UK, a 1 in 330 aqueous solution of phosphoric acid is approved as a disinfectant for foot-and-mouth disease. A specification for phosphoric acid is contained in the Food Chemicals Codex (FCC).(5) The EINECS number for phosphoric acid is 231-633-2. The PubChem Compound ID (CID) for phosphoric acid is 1004.