

Name: Propionic Acid
CAS No: Propionic acid [79-09-4]
USP-NF: Propionic Acid
Carboxyethane; ethanecarboxylic acid; E280; ethylformic acid; metacetonic acid; methylacetic acid; propanoic acid; pseudoacetic acid.
Propionic acid [79-09-4]
Propionic acid is primarily used as an antioxidant and antimicrobial preservative in foods, and in oral and topical pharmaceutical applications. It is also used as an esterifying agent.
Propionic acid occurs as a corrosive, oily liquid having a slightly pungent, disagreeable, rancid odor. It is flammable.
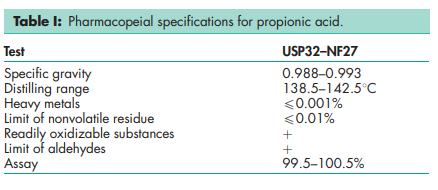
See Table I.
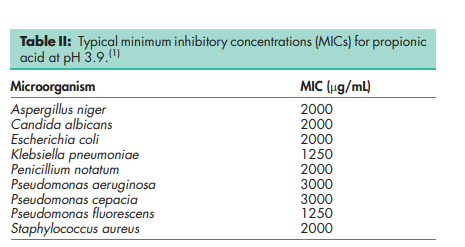
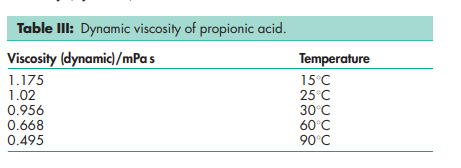
Antimicrobial activity see Table II. Autoignition temperature 9558C Boiling point 141.18C Dissociation constant pKa = 4.874 Flash point 52–588C (open cup) Melting point 21.58C Partition coefficients Octanol : water = 0.33. Refractive index nD 25 = 1.3848 Solubility Miscible with chloroform, ethanol (95%), ether, and water. Specific gravity 0.9934 Surface tension 27.21 mN/m (27.21 dynes/cm) at 158C Vapor density (relative) 2.56 (air = 1) Vapor pressure 320 Pa (2.4 mmHg) at 208C Viscosity (dynamic) see Table III
Although stable, propionic acid is flammable. It should be stored in an airtight container away from heat and flames.
Propionic acid is incompatible with alkalis, ammonia, amines, and halogens. It can be salted out of aqueous solutions by the addition of calcium chloride or other salts.
Propionic acid can be obtained from wood pulp waste liquor by fermentation. It can also be prepared from ethylene, carbon monoxide and steam; from ethanol and carbon monoxide using boron trifluoride catalyst; from natural gas; or as a by-product in the pyrolysis of wood. Very pure propionic acid can be obtained from propionitrile. Propionic acid can be found in dairy products in small amounts.
Propionic acid is generally regarded as a nontoxic and nonirritant material when used in low levels as an excipient. Up to 1% may be used in food applications (up to 0.3% in flour and cheese products). Propionic acid is readily metabolized. The pure form of propionic acid is corrosive and will cause burns to any area of contact. Both liquid and vapor forms are flammable. Concentrated propionic acid is harmful if swallowed, inhaled or absorbed through the skin. See also Sodium Propionate. LD50 (mouse, IV): 0.63 g/kg(2) LD50 (rabbit, skin): 0.5 g/kg LD50 (rat, oral): 2.6 g/kg
Propionic acid is corrosive and can cause eye and skin burns. It may be harmful if swallowed, inhaled or absorbed through the skin as a result of prolonged or widespread contact. Eye protection, PVC gloves, and suitable protective clothing should be worn. Propionic acid should be handled in a well-ventilated environment away from heat and flames. In the UK, the workplace exposure limits for propionic acid are 31 mg/m3 (10 ppm) long-term (8-hour TWA) and 46 mg/m3 (15 ppm) short-term.(3)
GRAS listed. Accepted for use in Europe as a food additive. In Japan, propionic acid is restricted to use as a flavoring agent
Calcium propionate; sodium propionate.
A specification for propionic acid is contained in the Food Chemicals Codex (FCC).(4) The EINECS number for propionic acid is 201-176-3. The PubChem Compound ID (CID) for propionic acid is 1032.