Name: Poloxamer
CAS No: a-Hydro-o-hydroxypoly(oxyethylene)poly(oxypropylene) poly (oxyethylene) block copolymer [9003-11-6]
BP: Poloxamers PhEur: Poloxamers USP-NF: Poloxamer
Lutrol; Monolan; Pluronic; poloxalkol; poloxamera; polyethylene– propylene glycol copolymer; polyoxyethylene–polyoxypropylene copolymer; Supronic; Synperonic.
a-Hydro-o-hydroxypoly(oxyethylene)poly(oxypropylene) poly (oxyethylene) block copolymer [9003-11-6]
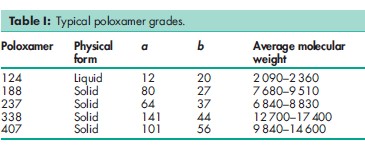
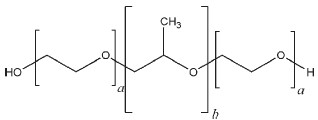
The poloxamer polyols are a series of closely related block copolymers of ethylene oxide and propylene oxide conforming to the general formula HO(C2H4O)a(C3H6O)b(C2H4O)aH. The grades included in the PhEur 6.0 and USP32–NF27 are shown in Table I. The PhEur 6.0 states that a suitable antioxidant may be added.
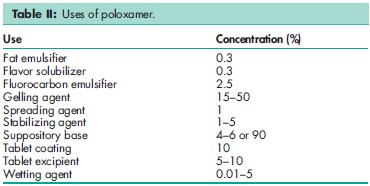
Poloxamers are nonionic polyoxyethylene–polyoxypropylene copolymers used primarily in pharmaceutical formulations as emulsifying or solubilizing agents.(1-8) The polyoxyethylene segment is hydrophilic while the polyoxypropylene segment is hydrophobic. All of the poloxamers are chemically similar in composition, differing only in the relative amounts of propylene and ethylene oxides added during manufacture. Their physical and surface-active properties vary over a wide range and a number of different types are commercially available; see Sections 4, 9, 10 and 18. Poloxamers are used as emulsifying agents in intravenous fat emulsions, and as solubilizing and stabilizing agents to maintain the clarity of elixirs and syrups. Poloxamers may also be used as wetting agents; in ointments, suppository bases, and gels; and as tablet binders and coatings. Poloxamer 188 has also been used as an emulsifying agent for fluorocarbons used as artificial blood substitutes, and in the preparation of solid-dispersion systems. More recently, poloxamers have found use in drug-delivery systems.(9–14) Therapeutically, poloxamer 188 is administered orally as a wetting agent and stool lubricant in the treatment of constipation; it is usually used in combination with a laxative such as danthron. Poloxamers may also be used therapeutically as wetting agents in eye-drop formulations, in the treatment of kidney stones, and as skin-wound cleansers. Poloxamer 338 and 407 are used in solutions for contact lens care. See Table II.
Poloxamers generally occur as white, waxy, free-flowing prilled granules, or as cast solids. They are practically odorless and tasteless. At room temperature, poloxamer 124 occurs as a colorless liquid.
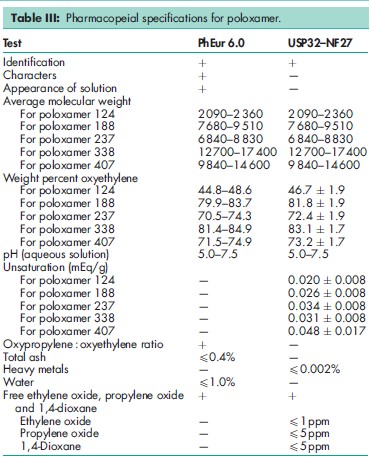
See Table III.
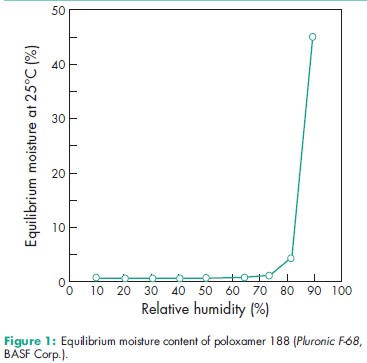
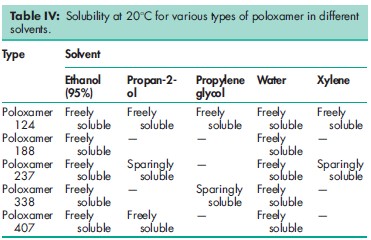
Acidity/alkalinity pH = 5.0–7.4 for a 2.5% w/v aqueous solution. Cloud point >1008C for a 1% w/v aqueous solution, and a 10% w/v aqueous solution of poloxamer 188. Density 1.06 g/cm3 at 258C Flash point 2608C Flowability Solid poloxamers are free flowing. HLB value 0.5–30; 29 for poloxamer 188. Melting point 168C for poloxamer 124; 52–578C for poloxamer 188; 498C for poloxamer 237; 578C for poloxamer 338; 52–578C for poloxamer 407. Moisture content Poloxamers generally contain less than 0.5% w/w water and are hygroscopic only at relative humidity greater than 80%. See also Figure 1. Solubility Solubility varies according to the poloxamer type; see also Table IV. Surface tension 19.8mN/m (19.8 dynes/cm) for a 0.1% w/v aqueous poloxamer 188 solution at 258C; 24.0mN/m (24.0 dynes/cm) for a 0.01% w/v aqueous poloxamer 188 solution at 258C; 26.0mN/m (26.0 dynes/cm) for a 0.001% w/v aqueous poloxamer solution at 258C. Viscosity (dynamic) 1000 mPa s (1000 cP) as a melt at 778C for poloxamer 188.
Poloxamers are stable materials. Aqueous solutions are stable in the presence of acids, alkalis, and metal ions. However, aqueous solutions support mold growth. The bulk material should be stored in a well-closed container in a cool, dry place.
Depending on the relative concentrations, poloxamer 188 is incompatible with phenols and parabens.
Poloxamer polymers are prepared by reacting propylene oxide with propylene glycol to form polyoxypropylene glycol. Ethylene oxide is then added to form the block copolymer.
Poloxamers are used in a variety of oral, parenteral, and topical pharmaceutical formulations, and are generally regarded as nontoxic and nonirritant materials. Poloxamers are not metabolized in the body. Animal toxicity studies, with dogs and rabbits, have shown poloxamers to be nonirritating and nonsensitizing when applied in 5% w/v and 10% w/v concentration to the eyes, gums, and skin. In a 14-day study of intravenous administration at concentrations up to 0.5 g/kg/day to rabbits, no overt adverse effects were noted. A similar study with dogs also showed no adverse effects at dosage levels up to 0.5 g/kg/day. In a longer-term study, rats fed 3% w/w or 5% w/w of poloxamer in food for up to 2 years did not exhibit any significant symptoms of toxicity. However, rats receiving 7.5% w/w of poloxamer in their diet showed some decrease in growth rate. No hemolysis of human blood cells was observed over 18 hours at 258C, with 0.001–10% w/v poloxamer solutions. Acute animal toxicity data for poloxamer 188:(15) LD50 (mouse, IV): 1 g/kg LD50 (mouse, oral): 15 g/kg LD50 (mouse, SC): 5.5 g/kg LD50 (rat, IV): 7.5 g/kg LD50 (rat, oral): 9.4 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended.
Included in the FDA Inactive Ingredients Database (IV injections; inhalations, ophthalmic preparations; oral powders, solutions, suspensions, and syrups; topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
—
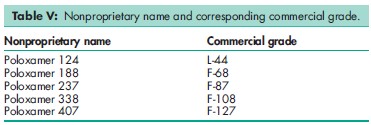
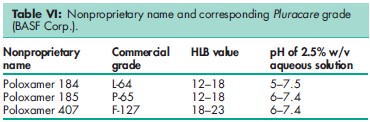
Although the USP32–NF27 contains specifications for five poloxamer grades, many more different poloxamers are commercially available that vary in their molecular weight and the proportion of oxyethylene present in the polymer. A series of poloxamers with greatly varying physical properties are thus available. The nonproprietary name ‘poloxamer’ is followed by a number, the first two digits of which, when multiplied by 100, correspond to the approximate average molecular weight of the polyoxypropylene portion of the copolymer and the third digit, when multiplied by 10, corresponds to the percentage by weight of the polyoxyethylene portion. Similarly, with many of the trade names used for poloxamers, e.g. Pluronic F-68 (BASF Corp.), the first digit arbitrarily represents the molecular weight of the polyoxypropylene portion and the second digit represents the weight percent of the oxyethylene portion. The letters ‘L’, ‘P’, and ‘F’, stand for the physical form of the poloxamer: liquid, paste, or flakes; see also Table V. Note that in the USA the trade name Pluronic is used by BASF Corp. for pharmaceutical-grade and industrial-grade poloxamers, while in Europe the trade name Lutrol is used by BASF Corp. for the pharmaceutical-grade material. Poloxamers for use in the cosmetic industry as oil-in-water emulsifiers, cleansers for mild facial products, and dispersing agents are marketed by BASF Corp. as Pluracare; the grades available are listed in Table VI. Studies on poloxamer 407, which shows thermoreversible properties for optimizing drug formulation temperature, have demonstrated immunomodulation and cytotoxicity promoting properties.(16) Poloxamer has been used in a poly(lactic-co-glycolic acid) (PLGA) : poloxamer and PLGA : poloxamine blend nanoparticle composition as novel carriers for gene delivery.(17) Specifications for poloxamer 331 and poloxamer 407 are contained in the Food Chemicals Codex (FCC).(18) The PubChem Compound ID (CID) for poloxamer is 24751.