

Name: Polyethylene Oxide
CAS No: Polyethylene oxide [25322-68-3]
USP-NF: Polyethylene Oxide
Polyox; polyoxiante; polyoxirane; polyoxyethylene.
Polyethylene oxide [25322-68-3]
See Table I.
The USP32–NF27 describes polyethylene oxide as a nonionic homopolymer of ethylene oxide, represented by the formula (CH2CH2O)n, where n represents the average number of oxyethylene groups. It may contain up to 3% of silicon dioxide or suitable antioxidant.
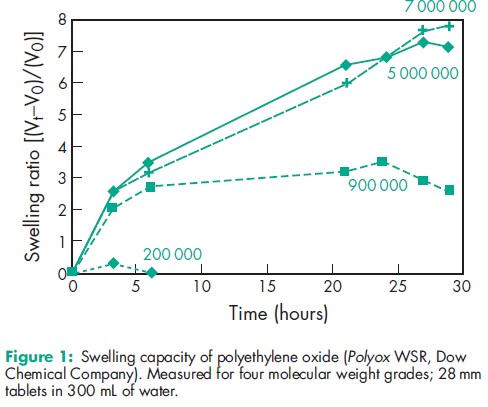
Polyethylene oxide can be used as a tablet binder at concentrations of 5–85%. The higher molecular weight grades provide delayed drug release via the hydrophilic matrix approach;(1,2) see Table I. Polyethylene oxide has also been shown to facilitate coarse extrusion for tableting(3) as well as being an aid in hot-melt extrusion.(4,5) The relationship between swelling capacity and molecular weight is a good guide when selecting products for use in immediate- or sustained-release matrix formulations; see Figure 1. Polyethylene oxide has been shown to be an excellent mucoadhesive polymer.(6) Low levels of polyethylene oxide are effective thickeners, although alcohol is usually added to waterbased formulations to provide improved viscosity stability; see Table II. Polyethylene oxide films demonstrate good lubricity when wet. This property has been utilized in the development of coatings for medical devices. Polyethylene oxide can be radiation crosslinked in solution to produce a hydrogel that can be used in wound care applications.

White to off-white, free-flowing powder. Slight ammoniacal odor.
See Table III.

Angle of repose 348 Density (true) 1.3 g/cm3 Melting point 65–708C Moisture content <1% NIR spectra see Figure 2. Solubility Polyethylene oxide is soluble in water and a number of common organic solvents such as acetonitrile, chloroform, and methylene chloride. It is insoluble in aliphatic hydrocarbons, ethylene glycol, and most alcohols.(7) Viscosity (dynamic) see Table II.
Store in tightly sealed containers in a cool, dry place. Avoid exposure to high temperatures since this can result in reduction in viscosity.
Polyethylene oxide is incompatible with strong oxidizing agents.
Polyethylene oxide is prepared by the polymerization of ethylene oxide using a suitable catalyst.(1)
Animal studies suggest that polyethylene oxide has a low level of toxicity regardless of the route of administration. It is poorly absorbed from the gastrointestinal tract but appears to be completely and rapidly eliminated. The resins are neither skin irritants nor sensitizers, and they do not cause eye irritation.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Polyethylene oxide may form an explosive dust–air mixture. Gloves, eye protection, a respirator, and other protective clothing should be worn.
Included in the FDA Inactive Ingredients Database (sustainedrelease tablets). Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Polyethylene glycol.
Polyethylene oxide and polyethylene glycol have the same CAS Registry Number 25322-68-3.