

Name: Povidone
CAS No: 1-Ethenyl-2-pyrrolidinone homopolymer [9003-39-8]
BP: Povidone JP: Povidone PhEur: Povidone USP: Povidone
E1201; Kollidon; Plasdone; poly[1-(2-oxo-1-pyrrolidinyl)ethylene]; polyvidone; polyvinylpyrrolidone; povidonum; Povipharm; PVP; 1- vinyl-2-pyrrolidinone polymer.
1-Ethenyl-2-pyrrolidinone homopolymer [9003-39-8]
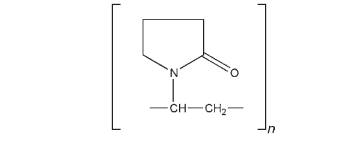
(C6H9NO)n 2500–3 000 000 The USP 32 describes povidone as a synthetic polymer consisting essentially of linear 1-vinyl-2-pyrrolidinone groups, the differing degree of polymerization of which results in polymers of various molecular weights. It is characterized by its viscosity in aqueous solution, relative to that of water, expressed as a K-value, in the range 10–120. The K-value is calculated using Fikentscher’s equation: where z is the relative viscosity of the solution of concentration c (in % w/v), and k is the K-value 10–3. Alternatively, the K-value may be determined from the following equation: where z is the relative viscosity of the solution of concentration c (in % w/v). Approximate molecular weights for different povidone grades are shown in Table I.
Although povidone is used in a variety of pharmaceutical formulations, it is primarily used in solid-dosage forms. In tableting, povidone solutions are used as binders in wet-granulation processes.(2,3) Povidone is also added to powder blends in the dry form and granulated in situ by the addition of water, alcohol, or hydroalcoholic solutions. Povidone is used as a solubilizer in oral and parenteral formulations, and has been shown to enhance dissolution of poorly soluble drugs from solid-dosage forms.(4–6) Povidone solutions may also be used as coating agents or as binders when coating active pharmaceutical ingredients on a support such as sugar beads. Povidone is additionally used as a suspending, stabilizing, or viscosity-increasing agent in a number of topical and oral suspensions and solutions. The solubility of a number of poorly soluble active drugs may be increased by mixing with povidone. See Table II. Special grades of pyrogen-free povidone are available and have been used in parenteral formulations;
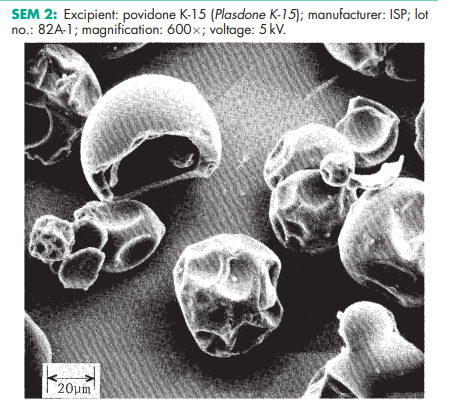
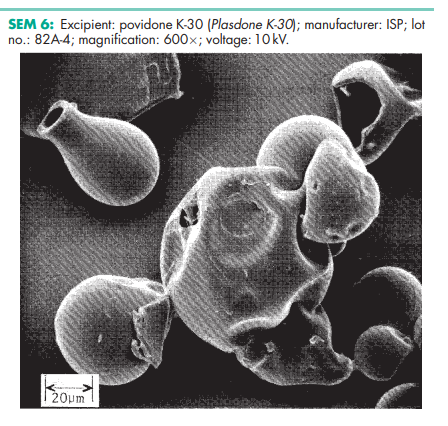
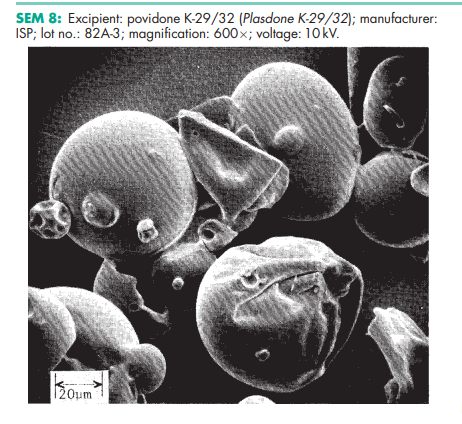
Povidone occurs as a fine, white to creamy-white colored, odorless or almost odorless, hygroscopic powder. Povidones with K-values equal to or lower than 30 are manufactured by spray-drying and occur as spheres. Povidone K-90 and higher K-value povidones are manufactured by drum drying and occur as plates.
See Table III.
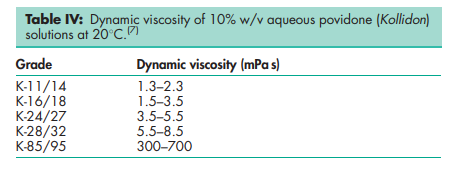
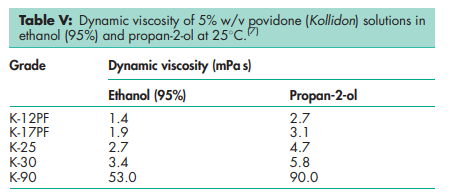
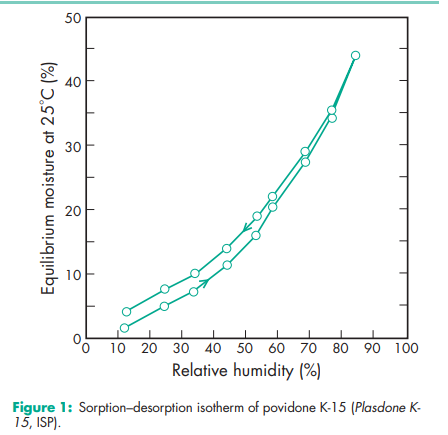
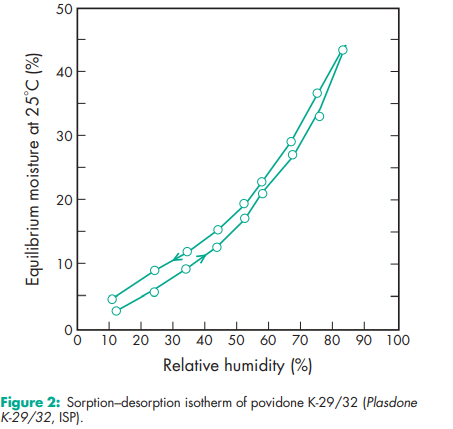
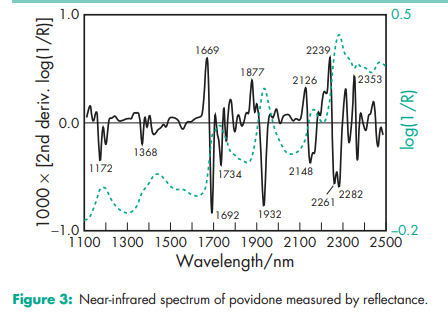
Acidity/alkalinity pH = 3.0–7.0 (5% w/v aqueous solution); pH = 4.0–7.0 (5% w/v aqueous solution) for Povipharm K90. Density (bulk) 0.29–0.39 g/cm3 for Plasdone. Density (tapped) 0.39–0.54 g/cm3 for Plasdone. Density (true) 1.180 g/cm3 Flowability 20 g/s for povidone K-15; 16 g/s for povidone K-29/32. Melting point Softens at 1508C. Moisture content Povidone is very hygroscopic, significant amounts of moisture being absorbed at low relative humidities. See Figures 1 and 2. NIR spectra see Figure 3. Particle size distribution Kollidon 25/30: 90% >50 mm, 50% >100 mm, 5% >200 mm; Kollidon 90: 90% >200 mm, 95% >250 mm.(7) Solubility Freely soluble in acids, chloroform, ethanol (95%), ketones, methanol, and water; practically insoluble in ether, hydrocarbons, and mineral oil. In water, the concentration of a solution is limited only by the viscosity of the resulting solution, which is a function of the K-value. Viscosity (dynamic) The viscosity of aqueous povidone solutions depends on both the concentration and the molecular weight of the polymer employed.


Povidone darkens to some extent on heating at 1508C, with a reduction in aqueous solubility. It is stable to a short cycle of heat exposure around 110–1308C; steam sterilization of an aqueous solution does not alter its properties. Aqueous solutions are susceptible to mold growth and consequently require the addition of suitable preservatives. Povidone may be stored under ordinary conditions without undergoing decomposition or degradation. However, since the powder is hygroscopic, it should be stored in an airtight container in a cool, dry place.
Povidone is compatible in solution with a wide range of inorganic salts, natural and synthetic resins, and other chemicals. It forms molecular adducts in solution with sulfathiazole, sodium salicylate, salicylic acid, phenobarbital, tannin, and other compounds; see Section 18. The efficacy of some preservatives, e.g. thimerosal, may be adversely affected by the formation of complexes with povidone.

Povidone is manufactured by the Reppe process. Acetylene and formaldehyde are reacted in the presence of a highly active copper acetylide catalyst to form butynediol, which is hydrogenated to butanediol and then cyclodehydrogenated to form butyrolactone. Pyrrolidone is produced by reacting butyrolactone with ammonia. This is followed by a vinylation reaction in which pyrrolidone and acetylene are reacted under pressure. The monomer, vinylpyrrolidone, is then polymerized in the presence of a combination of catalysts to produce povidone.
Povidone has been used in pharmaceutical formulations for many years, being first used in the 1940s as a plasma expander, although it has now been superseded for this purpose by dextran.(8) Povidone is widely used as an excipient, particularly in oral tablets and solutions. When consumed orally, povidone may be regarded as essentially nontoxic since it is not absorbed from the gastrointestinal tract or mucous membranes.(8) Povidone additionally has no irritant effect on the skin and causes no sensitization. Reports of adverse reactions to povidone primarily concern the formation of subcutaneous granulomas at the injection site of intramuscular injections formulated with povidone.(9) Evidence also exists that povidone may accumulate in the organs of the body following intramuscular injection.(10) A temporary acceptable daily intake for povidone has been set by the WHO at up to 25 mg/kg body-weight.(11) LD50 (mouse, IP): 12 g/kg(12) exists that povidone may accumulate in the organs of the body following intramuscular injection.(10) A temporary acceptable daily intake for povidone has been set by the WHO at up to 25 mg/kg body-weight.(11) LD50 (mouse, IP): 12 g/kg(12)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection, gloves, and a dust mask are recommended.
Accepted for use in Europe as a food additive. Included in the FDA Inactive Ingredients Database (IM and IV injections; ophthalmic preparations; oral capsules, drops, granules, suspensions, and tablets; sublingual tablets; topical and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Crospovidone
Povidone is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. The molecular adduct formation properties of povidone may be used advantageously in solutions, slow-release solid-dosage forms, and parenteral formulations. Perhaps the best-known example of povidone complex formation is povidone–iodine, which is used as a topical disinfectant. For accurate standardization of solutions, the water content of the solid povidone must be determined before use and taken into account for any calculations. Many excipients such as povidone may contain peroxides as trace contaminants. These can lead to degradation of an active pharmaceutical ingredient that is sensitive to oxidation. A specification for povidone is contained in the Food Chemicals Codex (FCC).(1