

Name: Polyoxyethylene Alkyl Ethers
CAS No: Polyethylene glycol monocetyl ether [9004-95-9], Polyethylene glycol monolauryl ether [9002-92-0], Polyethylene glycol monooleyl ether [9004-98-2], Polyethylene glycol monostearyl ether [9005-00-9]
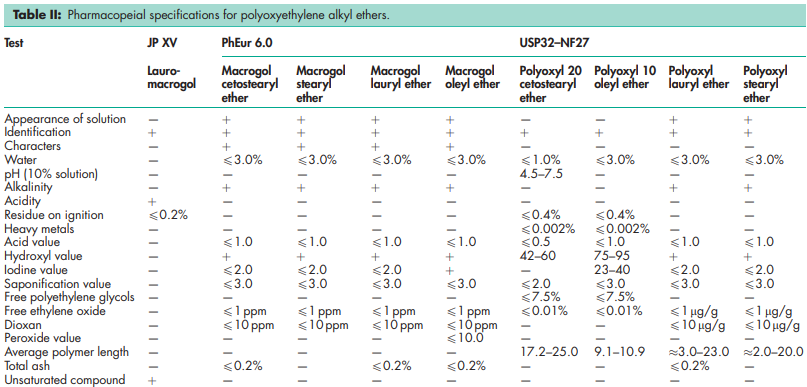
The polyoxyethylene alkyl ethers are a series of polyoxyethylene glycol ethers of n-alcohols (lauryl, oleyl, myristyl, cetyl, and stearyl alcohol). Of the large number of different materials commercially available, four types are listed in the USP32–NF27, one type in the JP XV, and four types in the PhEur 6.0. BP: Macrogol Cetostearyl Ether Macrogol Lauryl Ether Macrogol Oleyl Ether Macrogol Stearyl Ether JP: Lauromacrogol PhEur: Macrogol Cetostearyl Ether Macrogol Lauryl Ether Macrogol Oleyl Ether Macrogol Stearyl Ether USP-NF: Polyoxyl 20 Cetostearyl Ether Polyoxyl 10 Oleyl Ether Polyoxyl Lauryl Ether Polyoxyl Stearyl Ether Polyoxyethylene alkyl ethers are employed extensively in cosmetics, where the CTFA names laureth-N, myreth-N, ceteth-N, and steareth-N are commonly used. In this nomenclature, N is the number of ethylene oxide groups, e.g. steareth-20.
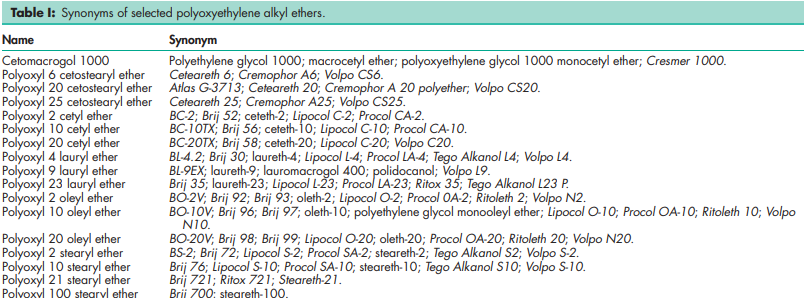
Polyoxyethylene alkyl ethers are nonionic surfactants produced by the polyethoxylation of linear fatty alcohols. Products tend to be mixtures of polymers of slightly varying molecular weights, and the numbers used to describe polymer lengths are average values. Two systems of nomenclature are used to describe these materials. The number ‘10’ in the name Volpo N10 refers to the approximate polymer length in oxyethylene units (i.e. y; see Section 5). The number ‘1000’ in the name ‘cetomacrogol 1000’ refers to the average molecular weight of the polymer chain. Synonyms applicable to polyoxyethylene alkyl ethers are shown below. Brij; Cremophor A; Cyclogol 1000; Empilan KB; Empilan KM; Emulgen; Ethosperse; Ethylan; macrogol ethers; macrogoli aether cetostearylicus; macrogoli aether laurilicus; macrogoli aether oleicus; macrogoli aether stearylicus; Marlowet; Plurafac; polyoxyethylene lauryl alcohol ether; Procol; Renex; Ritoleth; Ritox; Texofor A; Volpo. Table I shows synonyms for specific materials.
Polyethylene glycol monocetyl ether [9004-95-9], Polyethylene glycol monolauryl ether [9002-92-0], Polyethylene glycol monooleyl ether [9004-98-2], Polyethylene glycol monostearyl ether [9005-00-9]
See Sections 1, 2, and 5.
CH3(CH2)x(OCH2CH2)yOH In the formula, (x þ 1) is the number of carbon atoms in the alkyl chain, typically: 12 lauryl (dodecyl) 14 myristyl (tetradecyl) 16 cetyl (hexadecyl) 18 stearyl (octadecyl) and y is the number of ethylene oxide groups in the hydrophilic chain, typically 10–60. The polyoxyethylene alkyl ethers tend to be mixtures of polymers of slightly varying molecular weights, and the numbers quoted are average values. In cetomacrogol 1000, for example, x is 15 or 17, and y is 20–24
Polyoxyethylene alkyl ethers are nonionic surfactants widely used in topical pharmaceutical formulations and cosmetics, primarily as emulsifying agents for water-in-oil and oil-in-water emulsions, and the stabilization of microemulsions and multiple emulsions. Polyoxyethylene alkyl ethers are used as solubilizing agents for essential oils, perfumery chemicals, vitamin oils, and drugs of lowwater solubility such as cortisone acetate, griseofulvin, menadione,(1) chlordiazepoxide(2) and cholesterol.(3) They have applications as antidusting agents for powders; wetting and dispersing agents for coarse-particle liquid dispersions; and detergents, especially in shampoos, face washes and similar cosmetic cleaning preparations. They are used as gelling and foaming agents (e.g. Brij 72 gives a quick-breaking foam, while Brij 97 (15–20%), Volpo N series and Cremophor A25 (21–30%) give clear gels). Polyoxyethylene alkyl ethers have also been used in suppository formulations to increase the drug release from the suppository bases.(4–6) Polyoxyethylene alkyl ethers (especially laureth-23) have been used as a solubilizer and coating agent to provide hydrophilicity to polymeric nanoparticles.(7–9) Polyoxyethylene alkyl ethers such as polidocanol are suitable for use in injectable formulations as a solubilizer or dispersant.(10)
Polyoxyethylene alkyl ethers vary considerably in their physical appearance from liquids, to pastes, to solid waxy substances. They are colorless, white, cream-colored or pale yellow materials with a slight odor
See Table II.
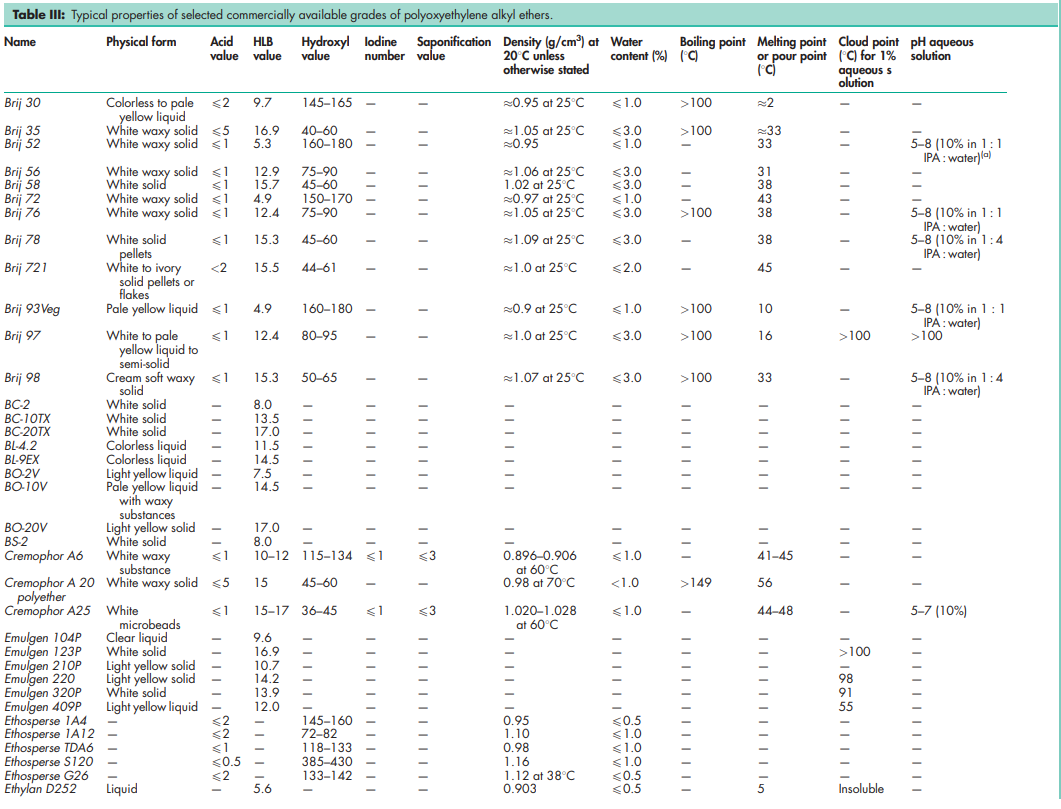
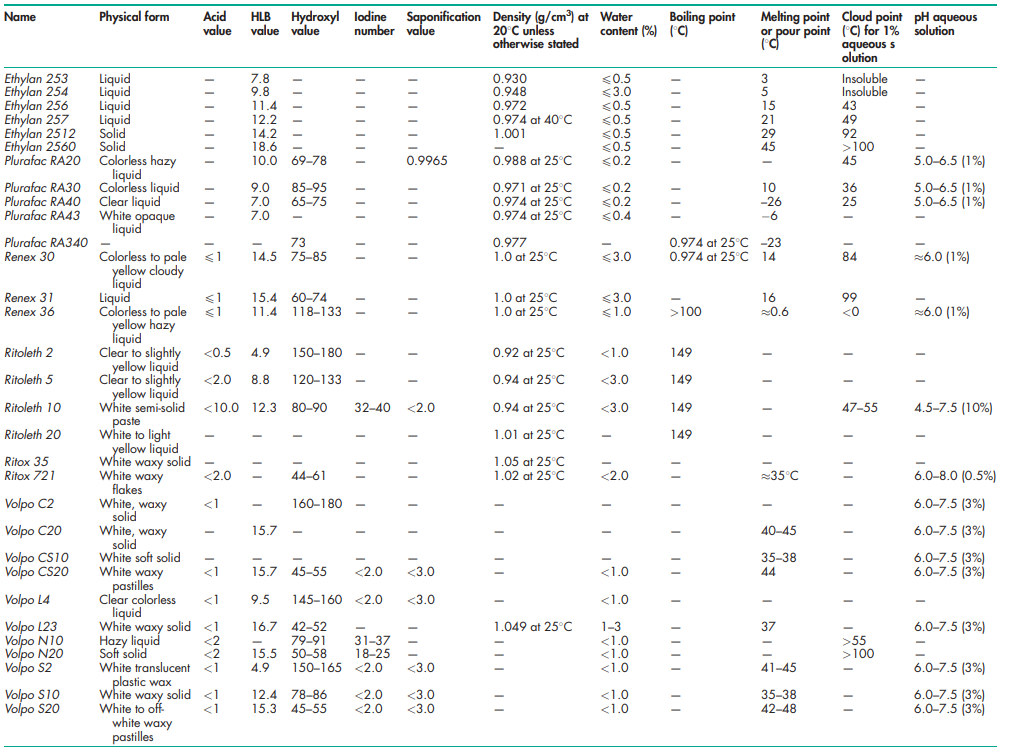
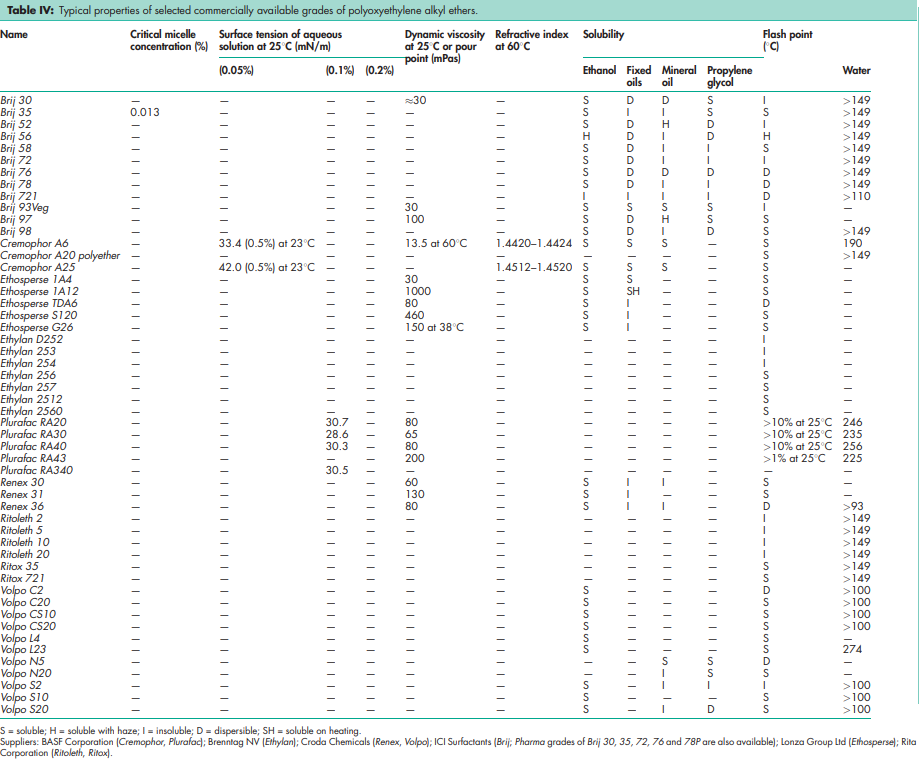
See Tables III and IV
Polyoxyethylene alkyl ethers are chemically stable in strongly acidic or alkaline conditions. The presence of strong electrolytes may, however, adversely affect the physical stability of emulsions containing polyoxyethylene alkyl ethers. On storage, polyoxyethylene alkyl ethers can undergo autoxidation, resulting in the formation of peroxides with an increase in acidity. Many commercially available grades are thus supplied with added antioxidants. Typically, a mixture of 0.01% butylated hydroxyanisole and 0.005% citric acid is used for this purpose. Polyoxyethylene alkyl ethers should be stored in an airtight container, in a cool, dry place.
Discoloration or precipitation may occur with iodides, mercury salts, phenolic substances, salicylates, sulfonamides, and tannins. Polyoxyethylene alkyl ethers are also incompatible with benzocaine, tretinoin(11) and oxidizable drugs.(12) The antimicrobial efficacy of some phenolic preservatives, such as the parabens, is reduced owing to hydrogen bonding. Cloud points are similarly depressed by phenols owing to hydrogen bonding between ether oxygen atoms and phenolic hydroxyl groups. Salts, other than nitrates, iodides, and thiocyanates (which cause an increase) can also depress cloud points.(13)
Polyoxyethylene alkyl ethers are prepared by the condensation of linear fatty alcohols with ethylene oxide. The reaction is controlled so that the required ether is formed with the polyethylene glycol of the desired molecular weight.
Polyoxyethylene alkyl ethers are used as nonionic surfactants in a variety of topical pharmaceutical formulations and cosmetics. The polyoxyethylene alkyl ethers form a series of materials with varying physical properties; manufacturers’ literature should be consulted for information on the applications and safety of specific materials. Although generally regarded as essentially nontoxic and nonirritant materials, some polyoxyethylene alkyl ethers, particularly when used in high concentration (>20%), appear to have a greater irritant potential than others. Animal toxicity studies suggest that polyoxyethylene alkyl ethers have a similar oral toxicity to other surfactants and can be regarded as being moderately toxic. Polyoxyethylene cetyl ether:(14) LD50 (mouse, oral): 2.60 g/kg LD50 (rabbit, skin): 40 g/kg/4 week intermittent LD50 (rat, oral): 2.50 g/kg Polyoxyethylene lauryl ether:(14) LD50 (mouse, IP): 0.16 g/kg LD50 (mouse, IV): 0.10 g/kg LD50 (mouse, oral): 4.94 g/kg LD50 (mouse, SC): 0.79 g/kg LD50 (rat, IV): 0.027 g/kg LD50 (rat, oral): 8.60 g/kg LD50 (rat, SC): 0.95 g/kg Polyoxyethylene oleyl ether:(14) LD50 (rat, oral): 25.8 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended.
Included in nonparenteral medicines licensed in the USA and UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Nonionic emulsifying wax.
Many other polyoxyethylene ethers are commercially available and are also used as surfactants. In addition to their surfactant properties, the series of polyoxyethylene ethers with lauryl side chains, e.g. nonoxynol 10, are also widely used as spermicides. Polyoxyethylene alkyl ethers have been studied in drug delivery systems containing oleosomes, hydrosomes, phosphosomes, vesicles(15) and niosomes.(16–18) An increased flux of estradiol niosomes through human stratum corneum in vitro has been demonstrated.(19) Polyoxyethylene alkyl ether niosomes encapsulating insulin have been investigated for oral drug delivery.(20) Polyoxyethylene alkyl ethers have been found to have an enhancing effect on the skin permeation of drugs such as ibuprofen,(21) methyl nicotinate,(22) and clotrimazole.(23) Enhanced ocular absorption of insulin from eye drops,(24) and an ocular insert device,(25) have been observed using polyoxyethylene alkyl ethers in the formulation systems. Increased buccal absorption of verapamil through porcine esophageal mucosa has also been reported.(26) A combination of a mucolytic agent possessing a free thiol group and a polyoxyethylene alkyl ether surfactant of similar polyoxyethylene and alkyl chain length have been reported to show effective enhancement in the intestinal absorption of poorly absorbed hydrophilic compounds.(27