

Name: Polyoxyethylene Castor Oil Derivatives
CAS No: Polyethoxylated castor oil [61791-12-6]
BP: Polyoxyl Castor Oil Hydrogenated Polyoxyl Castor Oil PhEur: Macrogolglycerol Ricinoleate Macrogolglycerol Hydroxystearate USP-NF: Polyoxyl 35 Castor Oil Polyoxyl 40 Hydrogenated Castor Oil Polyoxyethylene castor oil derivatives are a series of materials obtained by reacting varying amounts of ethylene oxide with either castor oil or hydrogenated castor oil. Several different types of material are commercially available, the best-known being the Cremophor series. Of these, two castor oil derivatives are listed in the PhEur 6.0 and USP32–NF27
Synonyms applicable to polyoxyethylene castor oil derivatives are shown below. See Table I for information on specific materials. Acconon; Arlatone; Cremophor; Etocas; Eumulgin; Jeechem; Lipocol; macrogolglyceroli hydroxystearas; macrogolglyceroli ricinoleas; Mapeg; Marlowet; Nikkol; Protachem; Simulsol.
Polyethoxylated castor oil [61791-12-6]
Polyoxyethylene castor oil derivatives are complex mixtures of various hydrophobic and hydrophilic components. Members within each range have different degrees of ethoxylation (moles)/PEG units as indicated by their numerical suffix (n). The chemical structures of the polyethoxylated hydrogenated castor oils are analogous to polyethoxylated castor oils with the exception that the double bond in the fatty chain has been saturated by hydrogenation. The PhEur 6.0 states that polyoxyl castor oil contains mainly ricinoleyl glycerol ethoxylated with 30–50 molecules of ethylene oxide (nominal value), with small amounts of macrogol ricinoleate, and of the corresponding free glycols. The PhEur 6.0 also states that polyoxyl hydrogenated castor oil contains mainly trihydroxystearyl glycerol ethoxylated with 7–60 molecules of ethylene oxide (nominal value). In polyoxyl 35 castor oil, the relatively hydrophobic constituents comprise about 83% of the total mixture, the main component being glycerol polyethylene glycol ricinoleate. Other hydrophobic constituents include fatty acid esters of polyethylene glycol along with some unchanged castor oil. The hydrophilic part (17%) consists of free polyethylene glycols and glycerol ethoxylates. Cremophor ELP, a ‘purified’ grade of Cremophor EL is also a polyoxyl 35 castor oil; it has a lower content of water (<0.5%), potassium (<15 ppm), and free fatty acids (C12–C18 <1%), particularly ricinoleic (<0.2%), oleic (<0.1%) and palmitic (<0.1%) acids, and hence is claimed to contribute to improved stability of some specific active ingredients. In polyoxyl 40 hydrogenated castor oil and polyoxyl 60 hydrogenated castor oil the main constituent is glycerol polyethylene glycol oxystearate, which together with fatty acid glycerol polyglycol esters, forms the hydrophobic constituent. The hydrophilic portion consists of polyethylene glycols and glycerol ethoxylate. Cremophor RH 410 is a mixture of 90% Cremophor RH 40 and 10% water. Cremophor CO 40, Cremophor 410 (90% Cremophore CO 40 þ 10% water), and Cremophor CO 455 (90% Cremophore CO 40 þ 5% water þ 5% propylene glycol) are cosmetic grades of polyoxyl hydrogenated castor oils.
See Section 4.
Polyoxyethylene castor oil derivatives are nonionic solubilizers and emulsifying agents used in oral, topical, and parenteral pharmaceutical formulations. Polyoxyl 35 castor oil is mainly used as an emulsifing and solubilizing agent, and is particularly suitable for the production of aqueous liquid preparations containing volatile oils, fat-soluble vitamins, and other hydrophobic substances.(1,2) Cremophor EL emulsifies or solubilizes the fat-soluble vitamins A, D, E, and K in aqueous solutions for oral and topical administration; in 1 mL of a 25% v/v aqueous solution it is possible to incorporate approximately 10 mg of vitamin A palmitate, approximately 10 mg of vitamin D, approximately 120 mg of vitamin E acetate, or approximately 120 mg of vitamin K1. In aqueous alcoholic solutions, it also very readily solubilizes essential oils. Aqueous solutions of hydrophobic drugs (e.g. miconazole, hexetidine, clotrimazole, benzocaine) can be prepared with Cremophor EL, which has also been used as a solubilizing agent for drugs like cyclosporin A,(3) paclitaxel,(4) and cisplatin.(5) Cremophor ELP is manufactured by purifying Cremophor EL and is therefore suitable for parenteral applications, e.g. Taxol preparations. In oral formulations, the taste of polyoxyl 35 castor oil can be masked by a banana flavor. Polyoxyl 35 castor oil has also been used as a solvent in proprietary injections of diazepam, propanidid, and alfaxalone with alfadolone acetate; see Section 14. A self-microemulsifying drug delivery system (SMEDDS) for oral bioavailability, and the enhancement of halofantrine,(6) and simvastatin,(7) have been prepared. Polyoxyl 35 castor oil has been used as a buffering agent for aqueous tropicamide eyedrops.(8) It has also been used in an aqueous mixture together with caprylic/capric glyceride for mucosal vaccination, providing a potential alternative to parenteral vaccination.(9) Polyoxyl 35 castor oil has been used to enhance the permeability of peptides across monolayers of Caco-2 cells by inhibiting the apically polarized efflux system, enhancing intestinal absorption of some drugs.(10) Cremophor has been used as a vehicle for boron neutron-capture therapy in mice, which is a form of radiation therapy used in the treatment of glioblastoma multiforme.(11) Polyoxyl 35 castor oil is also used in the production of glycerin suppositories. In veterinary practice, polyoxyl 35 castor oil can be used to emulsify cod liver oil, and oils and fats incorporated into animal feeding stuffs. Cremophor EL can enhance the bioavailability of substances such as vitamins in feed and veterinary medicines, improving their efficacy. In cosmetics, polyoxyl 35 castor oil is mainly used as a solubilizing agent for perfume bases and volatile oils in vehicles containing 30–50% v/v alcohol (ethanol or propan-2-ol). In hand lotions, it can be used to replace castor oil. Polyoxyl 40 hydrogenated castor oil may be used in preference to polyoxyl 35 castor oil in oral formulations as a solubilizer for fat soluble vitamins, essential oils and other hydrophobic pharmaceuticals. It has very little odor and it is almost tasteless. In aqueous alcoholic or completely aqueous solutions, polyoxyl 40 hydrogenated castor oil can be used to solubilize vitamins, essential oils, and certain drugs. Using 1 mL of a 25% v/v aqueous solution of polyoxyl 40 hydrogenated castor oil, it is possible to solubilize approximately 88 mg of vitamin A palmitate, or approximately 160 mg of vitamin A propionate. Other materials that can be solubilized are alfadolone, alfaxalone, anise oil, clotrimazole, diazepam, eucalyptol, gramicidin, hexachlorophene, hexetidine, levomepromazine, miconazole, propanidid, sage oil and thiopental. In aerosol vehicles that include water, the addition of polyoxyl 40 hydrogenated castor oil improves the solubility of the propellant in the aqueous phase. This enhancement applies both to dichlorodifluoromethane and to propane/butane mixtures. Foam formation in aqueous ethanol solutions containing polyoxyl 40 hydrogenated castor oil can be suppressed by the addition of small amounts of polypropylene glycol 2000. Polyoxyl 40 hydrogenated castor oil is also used as an emulsifier of fatty acids and alcohols. Polyoxyethylene castor oil derivatives have been used experimentally as a surfactant for the controlled release matrix pellet formulation containing nanocrystalline ketoprofen,(12) and for the transdermal delivery of vinpocetin.(13) Itraconazole has been incorporated in an aqueous parenteral formulation in an o/w microemulsion system containing polyoxyl 40 hydrogenated castor oil(14) and polyoxyl 50 hydrogenated castor oil.(15) A novel o/w microemulsion containing various emulsifiers including polyoxyl 40 hydrogenated castor oil was found to increase the solubility from 60 to 20 000 times of as many as nine poorly water soluble compounds, as well as to enhance the oral bioavailability of these compounds.(16) Hydrogenated castor oil (HCO) derivatives containing more than 20 oxyethylene units were found to prolong the plasma circulation times of menatetrenone incorporated in lipid emulsions.(17) Polyoxyl 60 hydrogenated castor oil derivatives have been reported to provide a self-microemulsifying system with enhanced oral absorption,(18) and a drastic reduction in plasma clearance of lipid emulsions.(19) It has been used in the formulation of liposomes,(20) and it has been suggested that more than 60% aids in the targeting of liposomes to the liver.(21) Polyoxyl 60 hydrogenated castor oil micellar solutions of cyclosporin A delivered the drug via the GI tract to the lymphatics with an extremely high selectivity.(22,23) Cremophor RH 40 and RH 60 have been used as additives to enhance the drug release from suppository formulations.(24,25)

Polyoxyl 35 castor oil occurs as a pale yellow, oily liquid that is clear at temperatures above 268C. It has a faint but characteristic odor and can be completely liquefied by heating to 268C. Polyoxyl 40 hydrogenated castor oil occurs as a white to yellowish, semisolid paste at 208C that liquefies at 308C. It has a very faint characteristic odor and is almost tasteless in aqueous solution. Polyoxyl 60 hydrogenated castor oil occurs as a white paste at room temperature. It has little taste or odor in aqueous solution
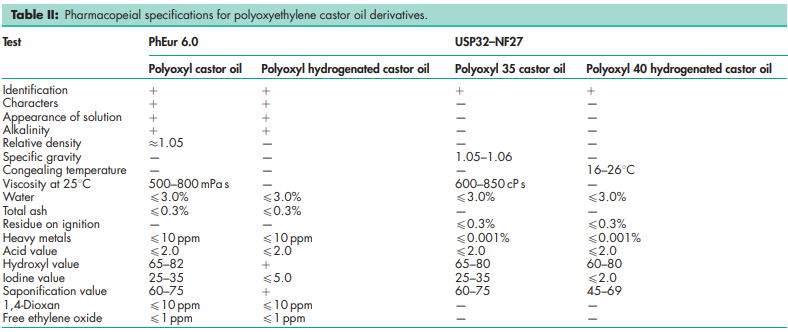
See Table II.
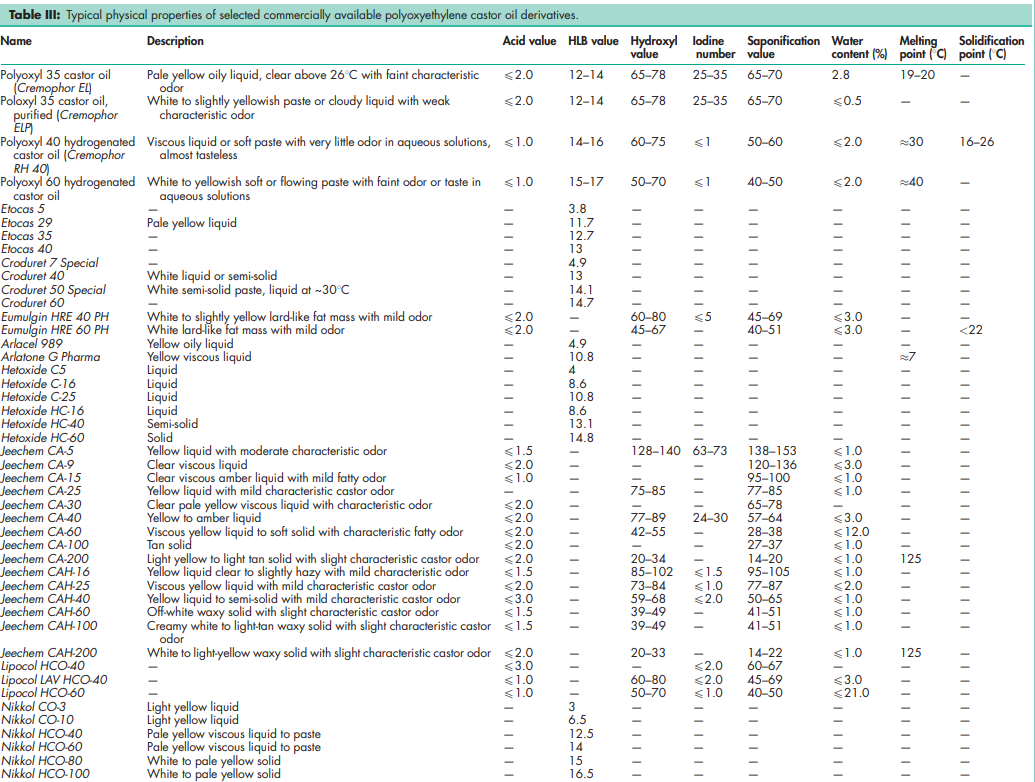
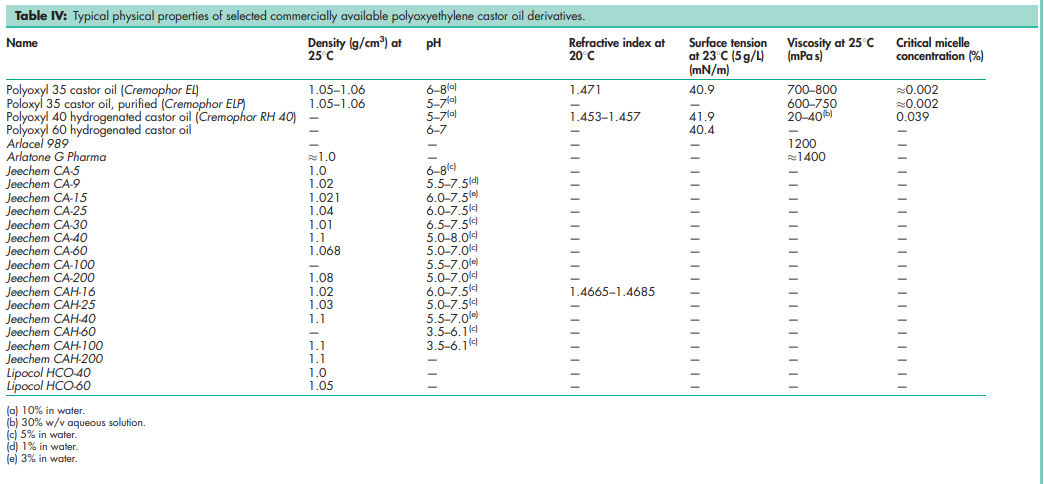
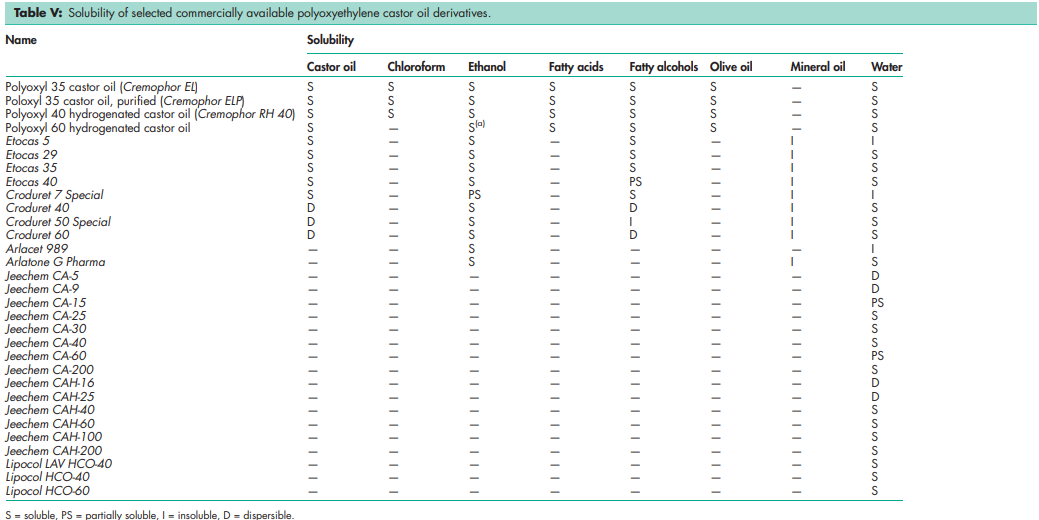
See Tables III, IV, and V.
Polyoxyl 35 castor oil forms stable solutions in many organic solvents such as chloroform, ethanol, and propan-2-ol; it also forms clear, stable, aqueous solutions. Polyoxyl 35 castor oil is miscible with other polyoxyethylene castor oil derivatives and on heating with fatty acids, fatty alcohols, and certain animal and vegetable oils. Solutions of polyoxyl 40 hydrogenated castor oil in aqueous alcohols and purely aqueous solutions are also stable. Solutions become cloudy as temperature increases. On heating of an aqueous solution, the solubility of polyoxyl 35 castor oil is reduced and the solution becomes turbid. Aqueous solutions of polyoxyl hydrogenated castor oil heated for prolonged periods may separate into solid and liquid phases on cooling. However, the product can be restored to its original form by homogenization. Aqueous solutions of polyoxyl 35 castor oil are stable in the presence of low concentrations of electrolytes such as acids or salts, with the exception of mercuric chloride; see Section 12. Heating together with very acidic or basic substances results in saponification. Aqueous solutions of polyoxyl 35 castor oil can be sterilized by autoclaving for 30 minutes at 1208C. In this process, a product may acquire a deeper color but this has no significance for product stability. Aqueous solutions of polyoxyl hydrogenated castor oil can similarly be sterilized by autoclaving at 1208C, but this may cause a slight decrease in the pH value. Phase separation may also be observed during sterilization, but can be remedied by agitating the solution while it is still hot. Although the method of manufacture used for polyoxyethylene castor oil derivatives ensures that they are near-sterile, microbial contamination can occur on storage. Polyoxyethylene castor oil derivatives should be stored in a wellfilled, airtight container, protected from light, in a cool, dry place. They are stable for at least 2 years if stored in the unopened original containers at room temperature (maximum 258C)
In strongly acidic or alkaline solutions, the ester components of polyoxyethylene hydrogenated castor oil are liable to saponify. In aqueous solution, polyoxyl 35 castor oil is stable toward most electrolytes in the concentrations normally employed. However, it is incompatible with mercuric chloride since precipitation occurs. Some organic substances may cause precipitation at certain concentrations, especially compounds containing phenolic hydroxyl groups, e.g. phenol, resorcinol, and tannins. Polyoxyl 40 hydrogenated castor oil and polyoxyl 60 hydrogenated castor oil are largely unaffected by the salts that cause hardness in water. Cremophor RH 40 was found to prolong the dissolution time of digoxin tablets.(26)
Polyoxyethylene castor oil derivatives are prepared by reacting varying amounts of ethylene oxide with either castor oil or hydrogenated castor oil under controlled conditions. Polyoxyl 35 castor oil is produced in this way by reacting 1 mole of castor oil with 35 moles of ethylene oxide. In the case of Cremophor ELP, this is followed by a purification process. Polyoxyl 40 hydrogenated castor oil is produced by reacting 1 mole of hydrogenated castor oil with 40–45 moles of ethylene oxide. Polyoxyl 60 hydrogenated castor oil is similarly produced by reacting 1 mole of hydrogenated castor oil with 60 moles of ethylene oxide.
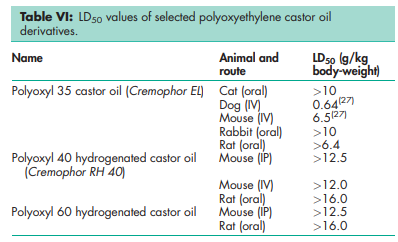
Polyoxyethylene castor oil derivatives are used in a variety of oral, topical, and parenteral pharmaceutical formulations. Acute and chronic toxicity tests in animals have shown polyoxyethylene castor oil derivatives to be essentially nontoxic and nonirritant materials; see Table VI.(27,28) However, there are reports of cardiovascular changes and nephrotoxicity in various species of animals.(29) Several serious anaphylactic reactions,(30–41) cardiotoxicity,(42–44) nephrotoxicity,(45,46) neurotoxicity,(47) and pulmonary toxicity(48) have also been observed in humans and animals following parenteral administration of formulations containing polyoxyethylene castor oil derivatives. The precise mechanism of the reaction is not known.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended.
Included in the FDA Inactive Ingredients Database (IV injections and ophthalmic solutions). Included in parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
Polyoxyethylene alkyl ethers; polyoxyethylene stearates.
Note that the trade name Cremophor (BASF Corp.) is also used for other polyoxyethylene derivatives e.g., the Cremophor A: series are polyoxyethylene alkyl ethers of cetostearyl alcohol. Polyoxyl 60 hydrogenated castor oil derivative has been investigated as an absorption enhancer in the absorption of erythropoietin from rat small intestine using gastrointestinal patches.(49) In another study, lipiodol and polyoxyl 60 hydrogenated castor oil derivative have been found to play an important role in the prolongation and selective retention of w/o emulsion or w/o/w multiple emulsion of doxorubicin hydrochloride in vitro and in vivo. (50)