

Name: Polyoxyethylene Stearates
CAS No: Polyethylene glycol stearate [9004-99-3], Polyethylene glycol distearate [9005-08-7]
The polyoxyethylene stearates are a series of polyethoxylated derivatives of stearic acid. Of the large number of different materials commercially available, one type is listed in the USP32–NF27. JP: Polyoxyl 40 Stearate USP-NF: Polyoxyl 40 Stearate
Ethoxylated fatty acid esters; macrogol stearates; Marlosol; PEG fatty acid esters; PEG stearates; polyethylene glycol stearates; poly(oxy-1,2-ethanediyl) a-hydro-o-hydroxyoctadecanoate; polyoxyethylene glycol stearates. Polyoxyethylene stearates are nonionic surfactants produced by polyethoxylation of stearic acid. Two systems of nomenclature are used for these materials. The number ‘8’ in the names ‘poloxyl 8 stearate’ or ‘polyoxyethylene 8 stearate’ refers to the approximate polymer length in oxyethylene units. The same material may also be designated ‘polyoxyethylene glycol 400 stearate’ or ‘macrogol stearate 400’ in which case, the number ‘400’ refers to the average molecular weight of the polymer chain. For synonyms applicable to specific polyoxyethylene stearates,
Polyethylene glycol stearate [9004-99-3], Polyethylene glycol distearate [9005-08-7]
See Table II.
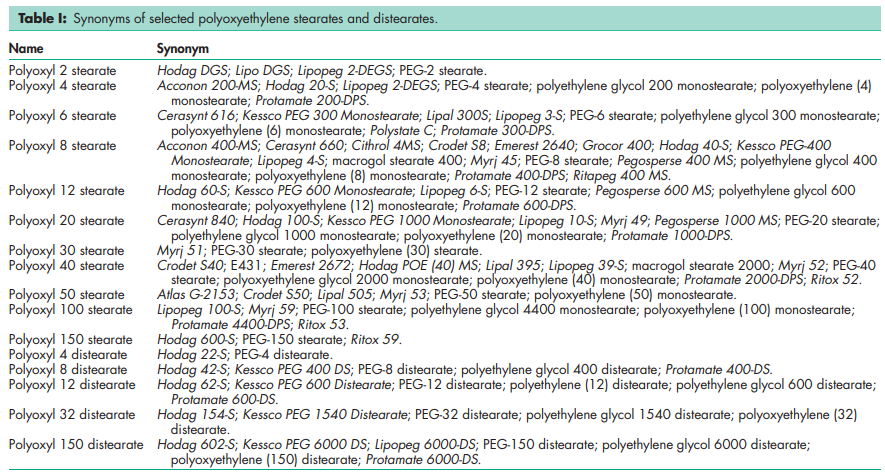
Structure A applies to the monostearate; where the average value of n is 6 for polyoxyl 6 stearate, 8 for polyoxyl 8 stearate, and so on. Structure B applies to the distearate; where the average value of n is 12 for polyoxyl 12 distearate, 32 for polyoxyl 32 distearate, and so on.
Polyoxyethylene stearates are generally used as emulsifiers in oil-inwater-type creams and lotions. Their hydrophilicity or lipophilicity depends on the number of ethylene oxide units present: the larger the number, the greater the hydrophilic properties. Polyoxyl 40 stearate has been used as an emulsifying agent in intravenous infusions.(1) Polyoxyethylene stearates are particularly useful as emulsifying agents when astringent salts or other strong electrolytes are present. They can also be blended with other surfactants to obtain any hydrophilic–lipophilic balance for lotions or ointment formulations. See Table III.
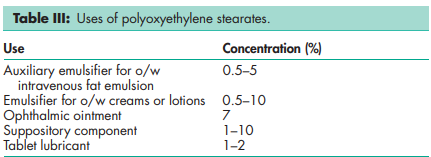
See Table IV.
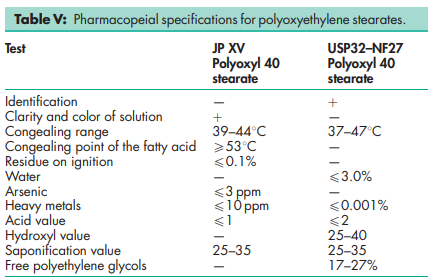
See Table V.
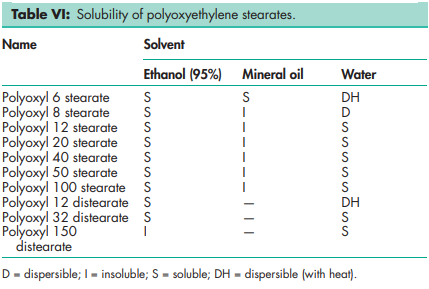
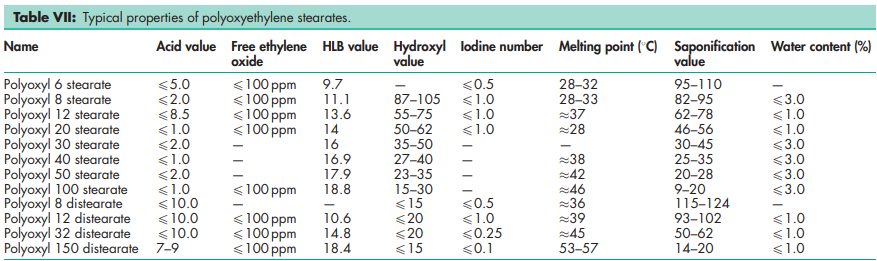
Flash point >1498C for poloxyl 8 stearate (Myrj 45). Solubility see Table VI. See also Table VII.
Polyoxyethylene stearates are generally stable in the presence of electrolytes and weak acids or bases. Strong acids and bases can cause gradual hydrolysis and saponification. The bulk material should be stored in a well-closed container, in a dry place, at room temperature.
Polyoxyethylene stearates are unstable in hot alkaline solutions owing to hydrolysis, and will also saponify with strong acids or bases. Discoloration or precipitation can occur with salicylates, phenolic substances, iodine salts, and salts of bismuth, silver, and tannins.(2–4) Complex formation with preservatives may also occur.(5) The antimicrobial activity of some materials such as bacitracin, chloramphenicol, phenoxymethylpenicillin, sodium penicillin, and tetracycline may be reduced in the presence of polyoxyethylene stearate concentrations greater than 5% w/w.(6,7
Polyoxyethylene stearates are prepared by the direct reaction of fatty acids, particularly stearic acid, with ethylene oxide.
Although polyoxyethylene stearates are primarily used as emulsifying agents in topical pharmaceutical formulations, certain materials, particularly polyoxyl 40 stearate, have also been used in intravenous injections and oral preparations.(1,4) Polyoxyethylene stearates have been tested extensively for toxicity in animals(8–13) and are widely used in pharmaceutical formulations and cosmetics. They are generally regarded as essentially nontoxic and nonirritant materials. Polyoxyl 8 stearate LD50 (hamster, oral): 27 g/kg LD50 (rat, oral): 64 g/kg Polyoxyl 20 stearate LD50 (mouse, IP): 0.2 g/kg LD50 (mouse, IV): 0.87 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Polyoxyethylene stearates that contain greater than 100 ppm of free ethylene oxide may present an explosion hazard when stored in a closed container. This is due to the release of ethylene oxide into the container headspace, where it can accumulate and so exceed the explosion limit
Included in the FDA Inactive Ingredients Database (dental solutions; IV injections; ophthalmic preparations; oral capsules and tablets; otic suspensions; topical creams, emulsions, lotions, ointments, and solutions; and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Polyethylene glycol; stearic acid.
It has been reported that polyoxyl 40 stearate may also enhance the activity of chemotherapeutic agents and reverse multidrug resistance of tumor cells.(14