

Name: Polyvinyl Acetate Phthalate
CAS No: Polyvinyl acetate phthalate [34481-48-6]
USP-NF: Polyvinyl Acetate Phthalate
Phthalavin; PVAP; Opaseal; Sureteric.
Polyvinyl acetate phthalate [34481-48-6]
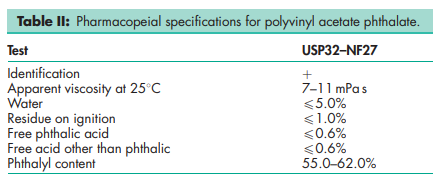
The USP32–NF27 describes polyvinyl acetate phthalate as a reaction product of phthalic anhydride and a partially hydrolyzed polyvinyl acetate. It contains not less than 55.0% and not more than 62.0% of phthalyl (o-carboxybenzoyl, C8H5O3) groups, calculated on an anhydrous acid-free basis. It has been reported that the free phthalic acid content is dependent on the source of the material.(1)
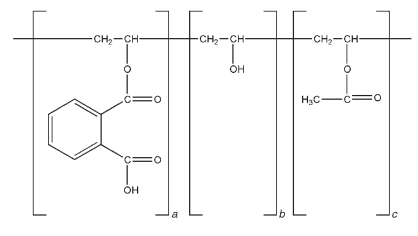
Depending on the phthalyl content, a will vary with b in mole percent. The acetyl content c remains constant depending on the starting material.
Polyvinyl acetate phthalate is a viscosity-modifying agent that is used in pharmaceutical formulations to produce enteric coatings for products and for the core sealing of tablets prior to a sugar-coating process. Polyvinyl acetate phthalate does not exhibit tackiness during coating and produces strong robust films. Plasticizers are often included in polyvinyl acetate phthalate coating formulations to enable a continuous, homogeneous, noncracking film to be produced. Polyvinyl acetate phthalate has been shown to be compatible with several plasticizers such as glyceryl triacetate, triethyl citrate, acetyl triethylcitrate, diethyl phthalate and polyethylene glycol 400. For enteric coating applications, polyvinyl acetate phthalate is dissolved in a solvent system together with other additives such as diethyl phthalate and stearic acid. Methanol may be used as the solvent if a colorless film is required; for a colored film, methanol or ethanol/water may be used depending on the amount of pigment to be incorporated. A weight increase of up to 8% is necessary for nonpigmented systems, whereas for pigmented systems a weight increase of 6% is usually required. A formulated, aqueous-based coating solution ( Sureteric, Colorcon) is available commercially for the enteric coating of tablets, hard and soft gelatin capsules and granules. More recently, hot-melt extrusion of coating polymers, such as polyvinyl acetate phthalate, has been described for the enteric coating of capsules.(2) Polyvinyl acetate phthalate has superseded materials such as shellac in producing the initial layers of coating (the sealing coat) in the sugar coating process for tablets. The sealing coating should be kept as thin as possible while providing an adequate barrier to moisture, a balance that is often difficult to achieve in practice. A solvent system containing a high proportion of industrial methylated spirits and other additives can be used. Two coats are usually sufficient to seal most tablets, although up to five may be necessary for tablets containing alkaline ingredients. If an enteric coating is also required, between six and 12 coats may be necessary; see Table I. The properties of polyvinyl acetate phthalate enteric coating have been compared with those of other enteric polymers such as cellulose acetate phthalate(3,4) and Eudragit L 30D. (4) The factors that affect the release kinetics from polyvinyl acetate phthalate enteric-coated tablets have also been described.(5) A method for enteric coating hypromellose capsules which avoids the sealing step prior to coating has been developed. The properties of several enteric coating polymers, including polyvinyl acetate phthalate, were assessed.(6)

Polyvinyl acetate phthalate is a free-flowing white to off-white powder and may have a slight odor of acetic acid. The material is essentially amorphous.(7)
See Table II.
The characteristics of polyvinyl acetate phthalate from two sources have been compared; values for molecular weight (60 700; 47 000), moisture content (3.74%; 2.20%) and density (1.31 g/cm3 ; 1.37 g/cm3 ) have been reported. The solubility of each polyvinyl acetate phthalate in a range of different solvents was described and scanning electron photomicrographs were produced to give evidence of the different polymer morphology.(8) Glass transition temperature A glass transition temperature of 42.58C has been reported for polyvinyl acetate phthalate; the glass transition temperature was shown to fall with the addition of increasing amounts of the plasticizer diethyl phthalate.(7) Solubility Soluble in ethanol and methanol; sparingly soluble in acetone and propan-2-ol; practically insoluble in chloroform, dichloromethane, and water. In buffer solutions, polyvinyl acetate phthalate (200 mg/L) is insoluble below pH 5 and becomes soluble at pH values above 5. Polyvinyl acetate pththalate shows a sharp solubility response with pH; this occurs at pH 4.5–5.0, which is lower than for most other polymers used for enteric coatings. Solubility is also influenced by ionic strength. See Table III.Viscosity (dynamic) The viscosity of a solution of polyvinyl acetate phthalate/methanol (1 : 1) is 5000 mPa s. In methanol/ dichloromethane systems, viscosity increases as the concentration of methanol in the system increases.

Polyvinyl acetate phthalate should be stored in airtight containers. It is relatively stable to temperature and humidity, and does not age, giving predictable release profiles even after prolonged storage. At high temperature and humidity, polyvinyl acetate phthalate undergoes less hydrolysis than other commonly used enteric coating polymers. In aqueous colloidal dispersions of polyvinyl acetate phthalate, the formation of free phthalic acid through hydrolysis was found to adversely affect physical stability.(1) Following storage at room temperature for 9 months, capsules coated with a commercial polyvinyl acetate phthalate formulation (Coateric) were found to retain gastroresistant properties and showed no apparent physical change; however, a delayed drug dissolution profile was observed after storage. Storage at 378C, or 378C and 80% relative humidity, for 3 months resulted in capsules having an unsatisfactory appearance.(4)
Polyvinyl acetate phthalate reacts with povidone to form an insoluble complex that precipitates out of solution;(9) benzocaine is also incompatible with polyvinyl acetate phthalate.(10) Erythromycin disperses in polyvinyl acetate phthalate and has been shown to be physically stable(11) while omeprazole exists in the amorphous form in polyvinyl acetate phthalate coatings with no evidence of interaction.(12)
Polyvinyl acetate phthalate is a reaction product of phthalic anhydride, sodium acetate, and a partially hydrolyzed polyvinyl alcohol. The polyvinyl alcohol is a low molecular weight grade, and 87–89 mole percent is hydrolyzed. Therefore, the polyvinyl acetate phthalate polymer is a partial esterification of a partially hydrolyzed polyvinyl acetate.
Polyvinyl acetate phthalate is used in oral pharmaceutical formulations and is generally regarded as an essentially nonirritant and nontoxic material when used as an excipient.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Gloves and eye protection are recommended.
Included in the FDA Inactive Ingredients Database (sustainedaction oral tablet). Included in nonparenteral medicines (enteric coated tablets; in printing ink formulations used for oral tablets and capsules) licensed in Europe. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Cellulose acetate phthalate; hypromellose phthalate; polymethacrylates; shellac
Polyvinyl acetate phthalate dissolves along the whole length of the duodenum.