

Name: Polyvinyl Alcohol
CAS No: Ethenol, homopolymer [9002-89-5]
PhEur: Poly(Vinyl Alcohol) USP: Polyvinyl Alcohol
Airvol; Alcotex; Celvol; Elvanol; Gelvatol; Gohsenol; Lemol; Mowiol; poly(alcohol vinylicus); Polyvinol; PVA; vinyl alcohol polymer.
Ethenol, homopolymer [9002-89-5]
(C2H4O)n 20 000–200 000 Polyvinyl alcohol is a water-soluble synthetic polymer represented by the formula (C2H4O)n. The value of n for commercially available materials lies between 500 and 5000, equivalent to a molecular weight range of approximately 20 000–200 000; see Table I.
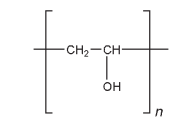
Polyvinyl alcohol is used primarily in topical pharmaceutical and ophthalmic formulations; see Table II.(1–3) It is used as a stabilizing agent for emulsions (0.25–3.0% w/v). Polyvinyl alcohol is also used as a viscosity-increasing agent for viscous formulations such as ophthalmic products. It is used in artificial tears and contact lens solutions for lubrication purposes, in sustained-release formulations for oral administration,(4) and in transdermal patches.(5) Polyvinyl alcohol may be made into microspheres when mixed with a glutaraldehyde solution.(6
Polyvinyl alcohol occurs as an odorless, white to cream-colored granular powder.
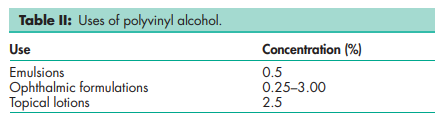
See Table III.
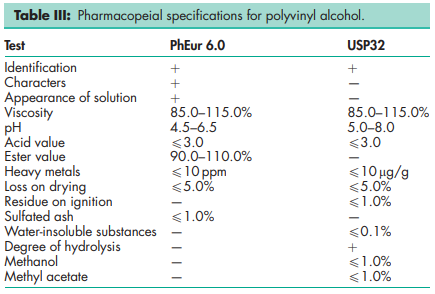
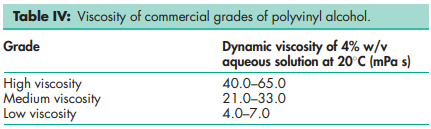
Melting point 2288C for fully hydrolyzed grades; 180–1908C for partially hydrolyzed grades. Refractive index nD 25 = 1.49–1.53 Solubility Soluble in water; slightly soluble in ethanol (95%); insoluble in organic solvents. Dissolution requires dispersion (wetting) of the solid in water at room temperature followed by heating the mixture to about 908C for approximately 5 minutes. Mixing should be continued while the heated solution is cooled to room temperature. Specific gravity 1.19–1.31 for solid at 258C; 1.02 for 10% w/v aqueous solution at 258C. Specific heat 1.67 J/g (0.4 cal/g) Viscosity (dynamic) see Table IV.
Polyvinyl alcohol is stable when stored in a tightly sealed container in a cool, dry place. Aqueous solutions are stable in corrosionresistant sealed containers. Preservatives may be added to the solution if extended storage is required. Polyvinyl alcohol undergoes slow degradation at 1008C and rapid degradation at 2008C; it is stable on exposure to light.
Polyvinyl alcohol undergoes reactions typical of a compound with secondary hydroxy groups, such as esterification. It decomposes in strong acids, and softens or dissolves in weak acids and alkalis. It is incompatible at high concentration with inorganic salts, especially sulfates and phosphates; precipitation of polyvinyl alcohol 5% w/v can be caused by phosphates. Gelling of polyvinyl alcohol solution may occur if borax is present.
Polyvinyl alcohol is produced through the hydrolysis of polyvinyl acetate. The repeating unit of vinyl alcohol is not used as the starting material because it cannot be obtained in the quantities and purity required for polymerization purposes. The hydrolysis proceeds rapidly in methanol, ethanol, or a mixture of alcohol and methyl acetate, using alkalis or mineral acids as catalysts.
Polyvinyl alcohol is generally considered a nontoxic material. It is nonirritant to the skin and eyes at concentrations up to 10%; concentrations up to 7% are used in cosmetics. Studies in rats have shown that polyvinyl alcohol 5% w/v aqueous solution injected subcutaneously can cause anemia and infiltrate various organs and tissues.(7) LD50 (mouse, oral): 14.7 g/kg LD50 (rat, oral): >20 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended. Polyvinyl alcohol dust may be an irritant on inhalation. Handle in a well-ventilated environment.
Included in the FDA Inactive Ingredients Database (ophthalmic preparations and oral tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Various grades of polyvinyl alcohol are commercially available. The degree of polymerization and the degree of hydrolysis are the two determinants of their physical properties. Pharmaceutical grades are partially hydrolyzed materials and are named according to a coding system. The first number following a trade name refers to the degree of hydrolysis and the second set of numbers indicates the approximate viscosity (dynamic), in mPa s, of a 4% w/v aqueous solution at 208C.