

Name: Potassium Bicarbonate
CAS No: Potassium bicarbonate [298-14-6]
BP: Potassium Bicarbonate PhEur: Potassium Hydrogen Carbonate USP: Potassium Bicarbonate
Carbonic acid monopotassium salt; E501; kalii hydrogenocarbonas; monopotassium carbonate; potassium acid carbonate; potassium hydrogen carbonate.
Potassium bicarbonate [298-14-6]
KHCO3 100.11
See Section 4.
As an excipient, potassium bicarbonate is generally used in formulations as a source of carbon dioxide in effervescent preparations, at concentrations of 25–50% w/w. It is of particular use in formulations where sodium bicarbonate is unsuitable, for example, when the presence of sodium ions in a formulation needs to be limited or is undesirable. Potassium bicarbonate is often formulated with citric acid or tartaric acid in effervescent tablets or granules; on contact with water, carbon dioxide is released through chemical reaction, and the product disintegrates. On occasion, the presence of potassium bicarbonate alone may be sufficient in tablet formulations, as reaction with gastric acid can be sufficient to cause effervescence and product disintegration. Potassium bicarbonate has also been investigated as a gasforming agent in alginate raft systems.(1,2) The effects of potassium bicarbonate on the stability and dissolution of paracetamol and ibuprofen have been described.(3) Potassium bicarbonate is also used in food applications as an alkali and a leavening agent, and is a component of baking powder. Therapeutically, potassium bicarbonate is used as an alternative to sodium bicarbonate in the treatment of certain types of metabolic acidosis. It is also used as an antacid to neutralize acid secretions in the gastrointestinal tract and as a potassium supplement.
Potassium bicarbonate occurs as colorless, transparent crystals or as a white granular or crystalline powder. It is odorless, with a saline or weakly alkaline taste.
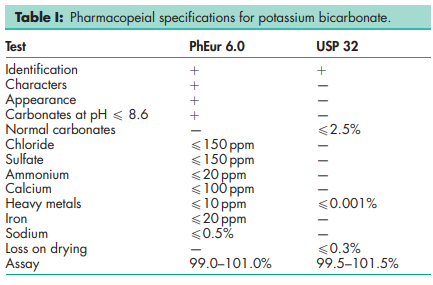
See Table I.
Acidity/alkalinity pH = 8.2 (for a 0.1 M aqueous solution); a 5% solution in water has a pH of 48.6. Solubility Soluble 1 in 4.5 of water at 08C, 1 in 2.8 of water at 208C, 1 in 2 of water at 508C; practically insoluble in ethanol (95%). Specific gravity 2.17
Potassium bicarbonate should be stored in a well-closed container in a cool, dry location. Potassium bicarbonate is stable in air at normal temperatures, but when heated to 100–2008C in the dry state, or in solution, it is gradually converted to potassium carbonate.
Potassium bicarbonate reacts with acids and acidic salts with the evolution of carbon dioxide.
Potassium bicarbonate can be made by passing carbon dioxide into a concentrated solution of potassium carbonate, or by exposing moist potassium carbonate to carbon dioxide, preferably under moderate pressure. Potassium bicarbonate also occurs naturally in the mineral calcinite.
Potassium bicarbonate is used in cosmetics, foods, and oral pharmaceutical formulations, where it is generally regarded as a relatively nontoxic and nonirritant material when used as an excipient. However, excessive consumption of potassium bicarbonate or other potassium salts may produce toxic manifestations of hyperkalemia.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended.
GRAS listed. Accepted as a food additive in Europe (the E number E501 refers to potassium carbonates). Included in nonparenteral medicines licensed in the UK and USA (chewable tablets; effervescent granules; effervescent tablets; lozenges; oral granules; oral suspensions; powder for oral solutions). Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Sodium bicarbonate.
One gram of potassium bicarbonate represents approximately 10 mmol of potassium and of bicarbonate; 2.56 g of potassium bicarbonate is approximately equivalent to 1 g of potassium. A specification for potassium bicarbonate is contained in the Food Chemicals Codex (FCC).(4) The EINECS number for potassium bicarbonate is 206-059-0. The PubChem Compound ID (CID) for potassium bicarbonate is 516893.