

Name: Butylparaben
CAS No: Butyl-4-hydroxybenzoate [94-26-8]
BP: Butyl Hydroxybenzoate JP: Butyl Parahydroxybenzoate PhEur: Butyl Parahydroxybenzoate USP-NF: Butylparaben
Butylis parahydroxybenzoas; butyl p-hydroxybenzoate; CoSept B; 4-hydroxybenzoic acid butyl ester; Lexgard B; Nipabutyl; Tegosept B; Trisept B; Uniphen P-23; Unisept B
Butyl-4-hydroxybenzoate [94-26-8]
C11H14O3 194.23
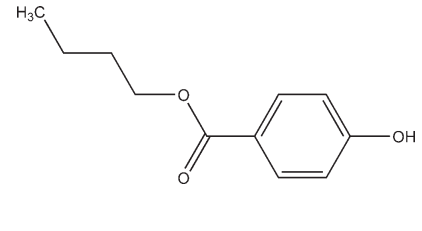
Butylparaben is widely used as an antimicrobial preservative in cosmetics and pharmaceutical formulations; see Table I. It may be used either alone or in combination with other paraben esters or with other antimicrobial agents. In cosmetics, it is the fourth most frequently used preservative.(1) As a group, the parabens are effective over a wide pH range and have a broad spectrum of antimicrobial activity, although they are most effective against yeasts and molds; see Section 10. Owing to the poor solubility of the parabens, paraben salts, particularly the sodium salt, are frequently used in formulations. However, this may raise the pH of poorly buffered formulations. See Methylparaben for further information.

Butylparaben occurs as colorless crystals or a white, crystalline, odorless or almost odorless, tasteless powder. SEM 1: Excipient: butylparaben; magnification: 240. SEM 2: Excipient: butylparaben; magnification: 2400


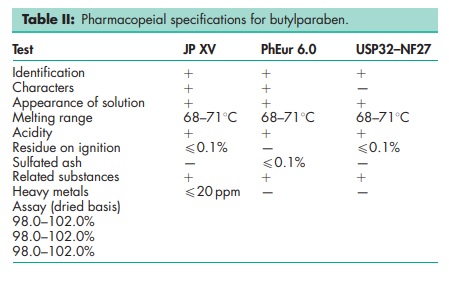
See Table II. See also Section 18.
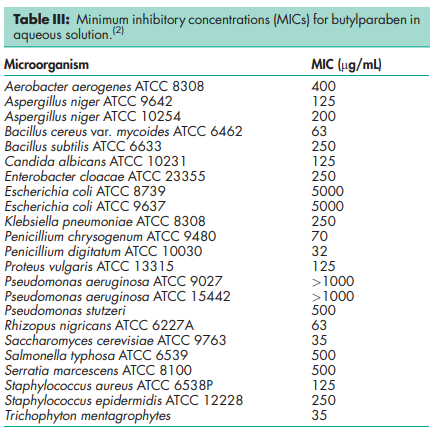
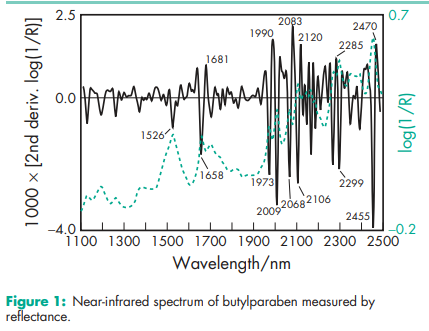
Antimicrobial activity Butylparaben exhibits antimicrobial activity between pH 4–8. Preservative efficacy decreases with increasing pH owing to the formation of the phenolate anion. Parabens are more active against yeasts and molds than against bacteria. They are also more active against Gram-positive than against Gram-negative bacteria; see Table III.(2) The activity of the parabens increases with increasing chain length of the alkyl moiety, but solubility decreases. Butylparaben is thus more active than methylparaben. Activity may be improved by using combinations of parabens since synergistic effects occur. Activity has also been reported to be improved by the addition of other excipients; see Methylparaben for further information Density (bulk) 0.731 g/cm3 Density (tapped) 0.819 g/cm3 Melting point 68–718C NIR spectra see Figure 1. Partition coefficients Values for different vegetable oils vary considerably and are affected by the purity of the oil; see Table IV.(3) Solubility see Table V.

Aqueous butylparaben solutions at pH 3–6 can be sterilized by autoclaving, without decomposition.(4) At pH 3–6, aqueous solutions are stable (less than 10% decomposition) for up to about 4 years at room temperature, while solutions at pH 8 or above are subject to rapid hydrolysis (10% or more after about 60 days at room temperature) Butylparaben should be stored in a well-closed container, in a cool, dry place.

The antimicrobial activity of butylparaben is considerably reduced in the presence of nonionic surfactants as a result of micellization.(6) Absorption of butylparaben by plastics has not been reported but appears probable given the behavior of other parabens. Some pigments, e.g. ultramarine blue and yellow iron oxide, absorb butylparaben and thus reduce its preservative properties.(7) Butylparaben is discolored in the presence of iron and is subject to hydrolysis by weak alkalis and strong acids. See also Methylparaben.
Butylparaben is prepared by esterification of p-hydroxybenzoic acid with n-butanol.
Butylparaben and other parabens are widely used as antimicrobial preservatives in cosmetics and oral and topical pharmaceutical formulations. Systemically, no adverse reactions to parabens have been reported, although they have been associated with hypersensitivity reactions generally appearing as contact dematitis. Immediate reactions with urticaria and bronchospasm have occurred rarely. See Methylparaben for further information. LD50 (mouse, IP): 0.23 g/kg(8) LD50 (mouse, oral): 13.2 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Butylparaben may be irritant to the skin, eyes, and mucous membranes, and should be handled in a well-ventilated environment. Eye protection, gloves, and a dust mask or respirator are recommended.
Included in the FDA Inactive Ingredients Database (injections; oral capsules, solutions, suspensions, syrups and tablets; rectal, and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Butylparaben sodium; ethylparaben; methylparaben; propylparaben. Butylparaben sodium Empirical formula C11H13NaO3 Molecular weight 216.23 CAS number [36457-20-2] Synonyms Butyl-4-hydroxybenzoate sodium salt; sodium butyl hydroxybenzoate. Appearance White, odorless or almost odorless, hygroscopic powder. Acidity/alkalinity pH = 9.5–10.5 (0.1% w/v aqueous solution) Solubility 1 in 10 of ethanol (95%); 1 in 1 of water. Comments Butylparaben sodium may be used instead of butylparaben because of its greater aqueous solubility. In unbuffered formulations, pH adjustment may be required.
Butylparaben is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. See Methylparaben for further information and references. The EINECS number for butylparaben is 202-318-7. The PubChem Compound ID (CID) for butylparaben is 7184.