

Name: Potassium Metabisulfite
CAS No: Dipotassium pyrosulfite [16731-55-8]
BP: Potassium Metabisulphite PhEur: Potassium Metabisulphite USP-NF: Potassium Metabisulfite
Disulfurous acid; dipotassium pyrosulfite; dipotassium salt; E224; kali disulfis; potassium pyrosulfite.
Dipotassium pyrosulfite [16731-55-8]
K2S2O5 222.32
See Section 4.
Potassium metabisulfite is used in applications similar to those of sodium metabisulfite in pharmaceuticals, and in the food, brewing, and wine making industries. It is used as an antioxidant, antimicrobial preservative and sterilizing agent.
Potassium metabisulfite occurs as white or colorless free-flowing crystals, crystalline powder, or granules, usually with an odor of sulfur dioxide.
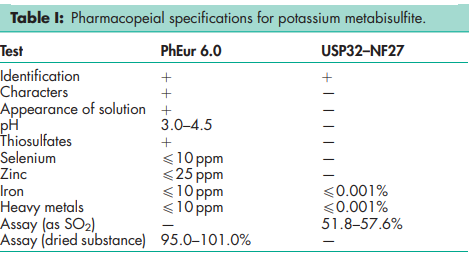
See Table I.
Acidity/alkalinity pH = 3.5–4.5 (5% w/v aqueous solution) Density (bulk) 1.1–1.3 g/cm3 Density (tapped) 1.2–1.5 g/cm3 Melting point 1908C although potassium metabisulfite decomposes at temperatures above 1508C. Solubility Soluble 1 in 2.2 of water; practically insoluble in ethanol (96%)
Potassium metabisulfite should be stored in a cool, dark place. When stored at a maximum temperature of 258C and maximum relative humidity of 45%, the shelf-life is 6 months. Potassium metabisulfite decomposes at temperatures above 1508C. In the air, it oxidizes to the sulfate, more readily in the presence of moisture. In aqueous solution, potassium metabisulfite forms potassium bisulfite (KHSO3) which exerts a strong reducing effect.
Potassium metabisulfite is incompatible with strong acids, water, and most common metals. It reacts with nitrites and sodium nitrate at room temperature, which occasionally results in the formation of flame. The reaction may be explosive if water is present. Potassium metabisulfite liberates SO2 with acids. Sulfites, including potassium metabisulfite, can react with various pharmaceutical compounds including sympathomimetics such as epinephrine (adrenaline),(1) chloramphenicol,(1) cisplatin,(2) and amino acids,(3) which can result in their pharmacological inactivation. Sulfites are also reported to react with phenylmercuric nitrate,(4,5) and may adsorb onto rubber closures.
Potassium metabisulfite is used in a variety of foods and pharmaceutical preparations, including oral, otic, rectal, and parenteral preparations. Potassium metabisulfite is considered a very irritating material, and may cause dermatitis on exposed skin.(6,7)Hypersensitivity reactions to potassium metabisulfite and other sulfites, mainly used as preservatives in food products, have been reported. Reactions include bronchospasm and anaphylaxis; some deaths have also been reported, especially in those with a history of asthma or atopic allergy.(8–12) These reactions have led to restrictions by the FDA on the use of sulfites in food applications.(13) However, this restriction has not been extended to their use in pharmaceutical applications. Indeed, epinephrine (adrenaline) injections used to treat severe allergic reactions may contain sulfites.(12,13) The WHO has set an acceptable daily intake of sulfites, as SO2, at up to 0.35 mg/kg body-weight.(14)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Protective gloves and safety goggles are recommended, and precautions should be taken to minimize exposure to the mucous membranes and respiratory tract. When heated to decomposition, it emits toxic fumes of SO2
GRAS listed. Accepted in Europe for use as a food additive in certain applications. Included in the FDA Inactive Ingredients Database (IM and IV injection; otic and rectal solutions and suspensions). Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Potassium bisulfite; sodium metabisulfite. Potassium bisulfite Empirical formula KHSO3 Molecular weight 120.2 CAS number [7773-03-7] Synonyms E228; potassium acid sulfite; potassium bisulphite; potassium hydrogen sulfite. Comments Accepted in Europe as a food additive in certain applications. Included in food and pharmaceutical applications similarly to potassium metabisulfite.
Like all sulfites, potassium metabisulfite is not recommended for use in foods that are a source of thiamin, owing to the instability of the vitamin in their presence. Such foods include meat, raw fruits and vegetables, fresh potatoes, and foods that are a source of vitamin B12. A specification for potassium metabisulfite is contained in the Food Chemicals Codex (FCC).(15) The EINECS number for potassium metabisulfite is 240-795-3. The PubChem Compound ID (CID) for potassium metabisulfite includes 28019 and 516928.