

Name: Ammonium Chloride
CAS No: Ammonium chloride [12125-02-9]
BP: Ammonium Chloride PhEur: Ammonium Chloride USP: Ammonium chloride
Ammonii chloridum; ammonium muriate; E510; sal ammoniac; salmiac.
Ammonium chloride [12125-02-9]
NH4Cl 53.49
See Section 4.
Ammonium chloride is used as an acidifying agent in oral formulations. It is also used as a food additive and antiseptic agent.(1) Ammonium chloride is used in the treatment of severe metabolic alkalosis to maintain the urine at an acid pH in the treatment of some urinary tract disorders or in forced acid diuresis.(2–4) It is also used as an expectorant in cough medicines.(5
Ammonium chloride occurs as colorless, odorless crystals or crystal masses. It is a white, granular powder with a cooling, saline taste. It is hygroscopic and has a tendency to cake.
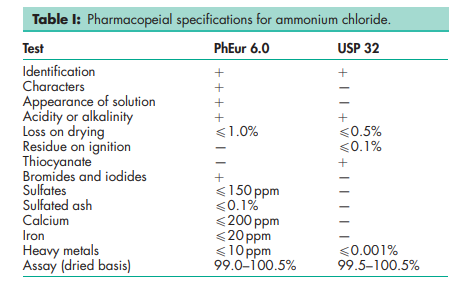
See Table I.
Acidity/alkalinity pH = 4.5–5.5 (5.5% w/w aqueous solutions at 258C) Density (bulk) 0.6–0.9 g/cm3 Hygroscopicity Hygroscopic with potential to cake. Melting point Decomposes at 3388C; sublimes without melting.(6) Solubility Soluble in water; hydrochloric acid and sodium chloride decrease its solubility in water. Also soluble in glycerin; sparingly soluble in methanol and ethanol. Almost insoluble in acetone, ether, and ethyl acetate. Specific gravity 1.527 g/cm3 Vapor pressure 133.3 Pa (1 mmHg) at 1608C
Ammonium chloride is chemically stable. It decomposes completely at 3388C to form ammonia and hydrochloric acid. Store in airtight containers in a cool, dry place.
Ammonium chloride is incompatible with strong acids and strong bases. It reacts violently with ammonium nitrate and potassium chlorate, causing fire and explosion hazards. It also attacks copper and its compounds.
Ammonium chloride is prepared commercially by reacting ammonia with hydrochloric acid.
Ammonium chloride is used in oral pharmaceutical formulations. The pure form of ammonium chloride is toxic by SC, IV, and IM routes, and moderately toxic by other routes. Potential symptoms of overexposure to fumes are irritation of eyes, skin, respiratory system: cough, dyspnea, and pulmonary sensitization.(7)Ammonium salts are an irritant to the gastric mucosa and may induce nausea and vomiting. LD50 (mouse, IP): 1.44 g/kg(8) LD50 (mouse, oral): 1.3 g/kg LD50 (rat, IM): 0.03 g/kg(9) LD50 (rat, oral): 1.65 g/kg(10)
Observe normal precautions appropriate to the circumstances and quantity of the material handled. All grades of ammonium chloride must be kept well away from nitrites and nitrates during transport and storage. They must be stored in a dry place, and effluent must not be discharged into the drains without prior treatment. Ammonium chloride decomposes on heating, producing toxic and irritating fumes (nitrogen oxides, ammonia, and hydrogen chloride).
GRAS listed. Included in the FDA Inactive Ingredients Database (oral syrup, tablets). Accepted for use as a food additive in Europe. Included in medicines licensed in the UK (eye drops; oral syrup).
Ammonia solution.
Ammonium chloride has the ability to cross the red blood cell membrane, and a solution that is isotonic to blood will still cause hemolytic rupture because it acts as a hypotonic solution. A specification for ammonium chloride is contained in the Food Chemicals Codex (FCC).(11) The EINECS number for ammonium chloride is 235-186-4. The PubChem Compound ID (CID) for ammonium chloride is 25517