

Name: Ascorbic Acid
CAS No: L-(þ)-Ascorbic acid [50-81-7]
BP: Ascorbic Acid JP: Ascorbic Acid PhEur: Ascorbic Acid USP: Ascorbic Acid
Acidum ascorbicum; C-97; cevitamic acid; 2,3-didehydro-L-threohexono-1,4-lactone; E300; 3-oxo-L-gulofuranolactone, enol form; vitamin C.
L-(þ)-Ascorbic acid [50-81-7]
C6H8O6 176.13
Ascorbic acid is used as an antioxidant in aqueous pharmaceutical formulations at a concentration of 0.01–0.1% w/v. Ascorbic acid has been used to adjust the pH of solutions for injection, and as an adjunct for oral liquids. It is also widely used in foods as an antioxidant. Ascorbic acid has also proven useful as a stabilizing agent in mixed micelles containing tetrazepam.(1)



Ascorbic acid occurs as a white to light-yellow-colored, nonhygroscopic, odorless, crystalline powder or colorless crystals with a sharp, acidic taste. It gradually darkens in color upon exposure to light.
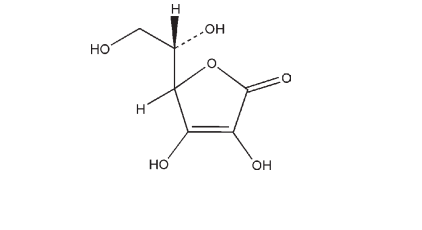
See Table I.
Acidity/alkalinity pH = 2.1–2.6 (5% w/v aqueous solution) Density (bulk) 0.7–0.9 g/cm3 for crystalline material; 0.5–0.7 g/cm3 for powder. Density (particle) 1.65 g/cm3 Density (tapped) 1.0–1.2 g/cm3 for crystalline material; 0.9–1.1 g/cm3 for powder. Density (true) 1.688 g/cm3 Dissociation constant pKa1 = 4.17; pKa2 = 11.57. Melting point 1908C (with decomposition) Moisture content 0.1% w/w
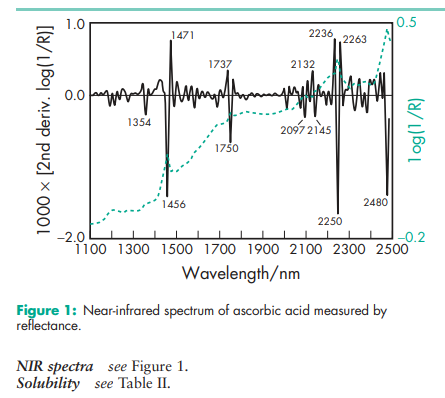
In powder form, ascorbic acid is relatively stable in air. In the absence of oxygen and other oxidizing agents it is also heat stable. Ascorbic acid is unstable in solution, especially alkaline solution, readily undergoing oxidation on exposure to the air.(2,3) The oxidation process is accelerated by light and heat and is catalyzed by traces of copper and iron. Ascorbic acid solutions exhibit maximum stability at about pH 5.4. Solutions may be sterilized by filtration. The bulk material should be stored in a well-closed nonmetallic container, protected from light, in a cool, dry place.
Incompatible with alkalis, heavy metal ions, especially copper and iron, oxidizing materials, methenamine, phenylephrine hydrochloride, pyrilamine maleate, salicylamide, sodium nitrite, sodium salicylate, theobromine salicylate, and picotamide.(4,5) Additionally, ascorbic acid has been found to interfere with certain colorimetric assays by reducing the intensity of the color produced.(6)
Ascorbic acid is prepared synthetically or extracted from various vegetable sources in which it occurs naturally, such as rose hips, blackcurrants, the juice of citrus fruits, and the ripe fruit of Capsicum annuum L. A common synthetic procedure involves the hydrogenation of D-glucose to D-sorbitol, followed by oxidation using Acetobacter suboxydans to form L-sorbose. A carboxyl group is then added at C1 by air oxidation of the diacetone derivative of Lsorbose and the resulting diacetone-2-keto-L-gulonic acid is converted to L-ascorbic acid by heating with hydrochloric acid
Ascorbic acid is an essential part of the human diet, with 40 mg being the recommended daily dose in the UK(7) and 60 mg in the USA.(8) However, these figures are controversial, with some advocating doses of 150 or 250 mg daily. Megadoses of 10 g daily have also been suggested to prevent illness although such large doses are now generally considered to be potentially harmful.(9–11) The body can absorb about 500 mg of ascorbic acid daily with any excess immediately excreted by the kidneys. Large doses may cause diarrhea or other gastrointestinal disturbances. Damage to the teeth has also been reported.(12) However, no adverse effects have been reported at the levels employed as an antioxidant in foods, beverages,(13) and pharmaceuticals. The WHO has set an acceptable daily intake of ascorbic acid, potassium ascorbate, and sodium ascorbate, as antioxidants in food, at up to 15 mg/kg bodyweight in addition to that naturally present in food.(14) LD50 (mouse, IV): 0.52 g/kg(15) LD50 (mouse, oral): 3.37 g/kg LD50 (rat, oral): 11.9 g/kg
Ascorbic acid may be harmful if ingested in large quantities and may be irritating to the eyes. Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and rubber or plastic gloves are recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (inhalations, injections, oral capsules, suspensions, tablets, topical preparations, and suppositories). Included in medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
Ascorbyl palmitate; erythorbic acid; sodium ascorbate
Many dosage forms for ascorbic acid have been developed for its administration to patients, including microencapsulation.(16) A specification for ascorbic acid is contained in the Food Chemicals Codex (FCC).(17) The EINECS number for ascorbic acid is 200-066-2. The PubChem Compound ID (CID) for ascorbic acid is 5785.