

Name: Ascorbyl Palmitate
CAS No: L-Ascorbic acid 6-hexadecanoate [137-66-6]
BP: Ascorbyl Palmitate PhEur: Ascorbyl Palmitate USP-NF: Ascorbyl Palmitate
L-Ascorbic acid 6-palmitate; ascorbylis palmitas; E304; 3-oxo-Lgulofuranolactone 6-palmitate; vitamin C palmitate.
L-Ascorbic acid 6-hexadecanoate [137-66-6]
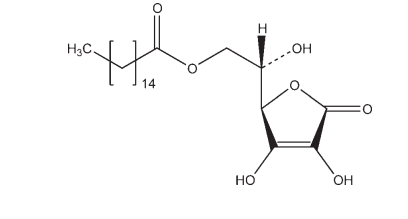
C22H38O7 414.54
Ascorbyl palmitate is primarily used either alone or in combination with alpha tocopherol as a stabilizer for oils in oral pharmaceutical formulations and food products; generally 0.05% w/v is used. It may also be used in oral and topical preparations as an antioxidant for drugs unstable to oxygen. The combination of ascorbyl palmitate with alpha tocopherol shows marked synergism, which increases the effect of the components and allows the amount used to be reduced. The solubility of ascorbyl palmitate in alcohol permits it to be used in nonaqueous and aqueous systems and emulsions.
Ascorbyl palmitate is a practically odorless, white to yellowish powder.
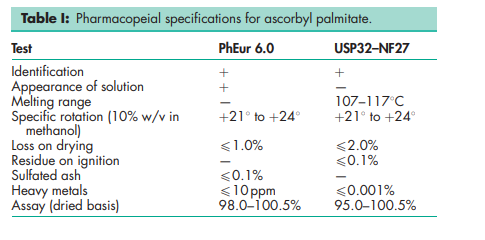
See Table I .
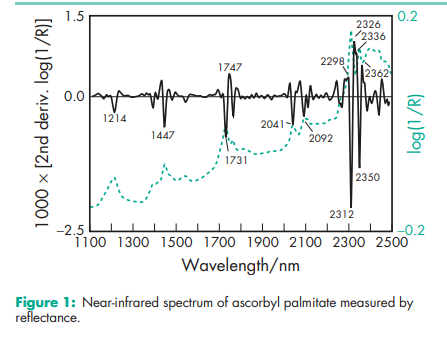
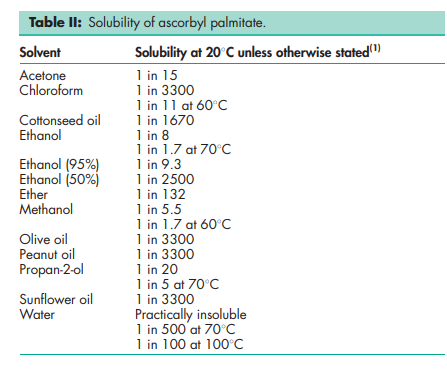
NIR spectra see Figure 1. Solubility see Table II.
Ascorbyl palmitate is stable in the dry state, but is gradually oxidized and becomes discolored when exposed to light and high humidity. In an unopened container, stored in a cool place, it has a shelf life of at least 12 months. During processing, temperatures greater than 658C should be avoided. The bulk material should be stored in an airtight container at 8–158C, protected from light.
Incompatibilities are known with oxidizing agents; e.g. in solution oxidation is catalyzed by trace metal ions such as Cu2þ and Fe3þ
Ascorbyl palmitate is prepared synthetically by the reaction of ascorbic acid with sulfuric acid followed by reesterification with palmitic acid.
Ascorbyl palmitate is used in oral pharmaceutical formulations and food products, and is generally regarded as an essentially nontoxic and nonirritant material. The WHO has set an estimated acceptable daily intake for ascorbyl palmitate at up to 1.25 mg/kg bodyweight.(2) LD50 (mouse, oral): 25 g/kg(3) LD50 (rat, oral): 10 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Ascorbyl palmitate dust may cause irritation to the eyes and respiratory tract. Eye protection is recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral, rectal, topical preparations). Included in nonparenteral medicines licensed in the UK.
Ascorbic acid; sodium ascorbate.
In order to maximize the stability and efficacy of ascorbyl palmitate the following precautions are recommended: stainless steel, enamel, or glass should be used; deaeration (vacuum) procedures and inert gas treatment are recommended where feasible; protect from light and radiant energy. The formation of ascorbyl palmitate vesicles (Aspasomes) and their pharmaceutical applications has been investigated.(4) The EINECS number for ascorbyl palmitate is 205-305-4. The PubChem Compound ID (CID) for ascorbyl palmitate is 5282566.