

Name: Acesulfame Potassium
CAS No: 6-Methyl-1,2,3-oxathiazin-4(3H)-one-2,2-dioxide potassium salt [55589-62-3]
BP: Acesulfame Potassium PhEur: Acesulfame Potassium USP-NF: Acesulfame Potassium
Acesulfame K; ace K; acesulfamum kalicum; E950; 6-methyl-3,4 dihydro-1,2,3-oxathiazin-4(3H)-one-2,2-dioxide potassium salt; potassium 6-methyl-2,2-dioxo-oxathiazin-4-olate; Sunett; Sweet One
6-Methyl-1,2,3-oxathiazin-4(3H)-one-2,2-dioxide potassium salt [55589-62-3]
C4H4KNO4S 201.24
Acesulfame potassium is used as an intense sweetening agent in cosmetics, foods, beverage products, table-top sweeteners, vitamin and pharmaceutical preparations, including powder mixes, tablets, and liquid products. It is widely used as a sugar substitute in compounded formulations,(1) and as a toothpaste sweetener.(2) The approximate sweetening power is 180–200 times that of sucrose, similar to aspartame, about one-third as sweet as sucralose, one-half as sweet as sodium saccharin, and about 4-5 times sweeter than sodium cyclamate.(3) It enhances flavor systems and can be used to mask some unpleasant taste characteristics.
Acesulfame potassium occurs as a colorless to white-colored, odorless, crystalline powder with an intensely sweet taste.
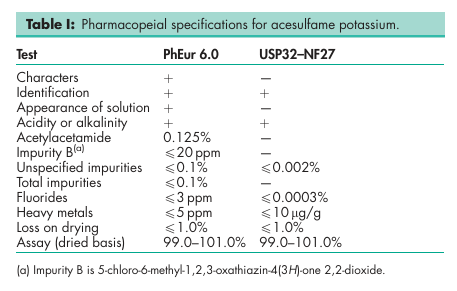
See Table I.
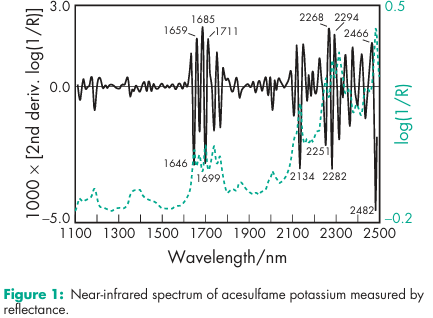
Acidity/alkalinity pH = 5.5–7.5 (1% w/v aqueous solution) Bonding index 0.007(4) Brittle fracture index 0.08(4) Density (bulk) 1.04g/cm3 (4) Density (tapped) 1.28g/cm3 (4) Elastic modulus 4000MPa(4) Flowability 19% (Carr compressibility index)(4) Melting point 2508C NIR spectra see Figure 1. Solubility see Table II.


Acesulfamepotassiumpossessesgoodstability. Inthebulkformit showsnosignofdecompositionatambienttemperatureovermany years. Inaqueoussolutions(pH3.0–3.5at208C)noreductionin sweetnesswasobservedoveraperiodofapproximately2years. Stabilityatelevatedtemperatures isgood,althoughsomedecom positionwasnotedfollowingstorageat408Cforseveralmonths. Sterilizationandpasteurizationdonotaffectthetasteofacesulfame potassium.(7) Thebulkmaterialshouldbestoredinawell-closedcontainerina cool,dryplaceandprotectedfromlight.
Acesulfame potassiumis synthesized fromacetoacetic acid tert butylesterandfluorosulfonyl isocyanate.Theresultingcompound is transformedtofluorosulfonyl acetoaceticacidamide,whichis thencyclizedinthepresenceofpotassiumhydroxidetoformthe oxathiazinonedioxideringsystem.Becauseofthestrongacidityof thiscompound, thepotassiumsalt isproduceddirectly.(8) Analternativesynthesis route foracesulfamepotassiumstarts withthereactionbetweendiketeneandamidosulfonicacid. Inthe presence of dehydrating agents, and after neutralizationwith potassiumhydroxide,acesulfamepotassiumisformed.
Acesulfamepotassiumiswidelyusedinbeverages,cosmetics,foods, andpharmaceutical formulations, and isgenerallyregardedasa relatively nontoxic and nonirritant material. Pharmacokinetic studieshaveshownthatacesulfamepotassiumisnotmetabolized andisrapidlyexcretedunchangedintheurine.Long-termfeeding studiesinratsanddogsshowednoevidencetosuggestacesulfame potassiumismutagenicorcarcinogenic.(9) TheWHOhas set anacceptabledaily intake for acesulfame potassium of up to 15mg/kg body-weight.(9) The Scientific CommitteeforFoodsoftheEuropeanUnionhassetadailyintake valueofupto9mg/kgofbody-weight.(3) LD50(rat, IP):2.2g/kg(7) LD50(rat,oral):6.9–8.0g/kg
Observenormalprecautionsappropriatetothecircumstancesand quantityofmaterial handled.Eyeprotection, gloves, andadust maskarerecommended.
Included in theFDAInactive IngredientsDatabase fororal and sublingualpreparations. IncludedintheCanadianListofAccep tableNon-medicinal Ingredients.Acceptedforuse inEuropeasa foodadditive.Itisalsoacceptedforuseincertainfoodproductsin theUSAandseveral countries inCentralandSouthAmerica, the MiddleEast,Africa,Asia,andAustralia.
Alitame.
Theperceivedintensityof sweeteners relative tosucrosedepends upontheirconcentration, temperatureof tasting,andpH,andon theflavorandtextureoftheproductconcerned. Intensesweeteningagentswillnotreplacethebulk, textural,or preservative characteristics of sugar, if sugar is removed froma formulation. Synergistic effects for combinations of sweeteners have been reported, e.g. acesulfame potassiumwith aspartame or sodium cyclamate; seealsoAspartame.Aternarycombinationof sweet enersthatincludesacesulfamepotassiumandsodiumsaccharinhas agreaterdecrease insweetnessuponrepeatedtastingthanother combinations.(10) Note that free acesulfame acid is not suitable for use as a sweetener. Aspecification for acesulfamepotassiumis contained in the FoodChemicalsCodex(FCC).(11)