

Name: Dextrates
CAS No: Dextrates [39404-33-6]
USP-NF: Dextrates
Candex; Emdex.
Dextrates [39404-33-6]
The USP32–NF27 describes dextrates as a purified mixture of saccharides resulting from the controlled enzymatic hydrolysis of starch. It may be either hydrated or anhydrous. Its dextrose equivalent is not less than 93.0% and not more than 99.0%, calculated on the dried basis.
See Section 4.
Dextrates is a directly compressible tablet diluent used in chewable, nonchewable, soluble, dispersible, and effervescent tablets.(1–3) It is a free-flowing material and glidants are thus unnecessary. Lubrication with magnesium stearate (0.5–1.0% w/w) is recommended.(4) Dextrates may also be used as a binding agent by the addition of water, no further binder being required.(4) Tablets made from dextrates increase in crushing strength in the first few hours after manufacture, but no further increase occurs on storage.(5)
Dextrates is a purified mixture of saccharides resulting from the controlled enzymatic hydrolysis of starch. It is either anhydrous or hydrated. In addition to dextrose, dextrates contains 3–5% w/w maltose and higher polysaccharides. Dextrates comprises white spray-crystallized free-flowing porous spheres. It is odorless with a sweet taste (about half as sweet as sucrose).
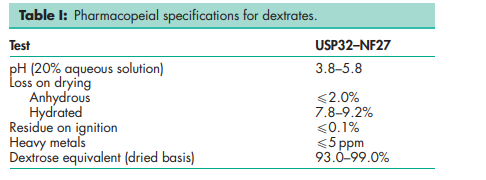
See Table I.
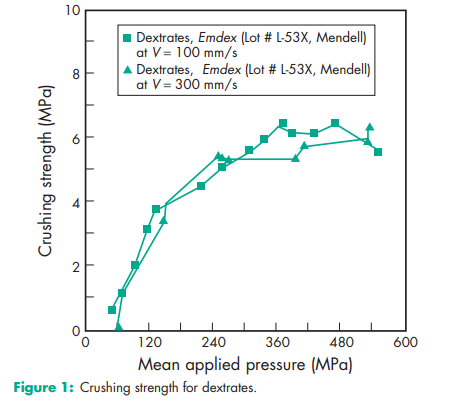
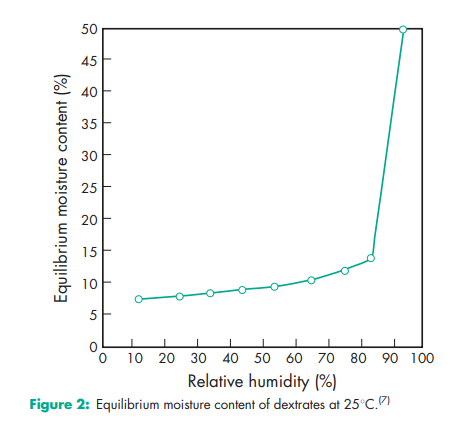
Angle of repose 26.48 (6) Compressibility see Figure 1.(6) Density (bulk) 0.68 g/cm3 (6) Density (tapped) 0.72 g/cm3 (6) Density (true) 1.539 g/cm3 Hausner ratio 1.05 Flowability 9.3 g/s (6) Heat of combustion 16.8–18.8 J/g (4.0–4.5 cal/g) Heat of solution 105 J/g (–25 cal/g) Melting point 1418C Moisture content 7.8–9.2% w/w (hydrated form). See also Figure 2.(7) NIR spectra see Figure 3. Particle size distribution Not more than 3% retained on a 840 mm sieve; not more than 25% passes through a 150 mm sieve. Mean particle size 190–220 mm. Solubility Soluble 1 in 1 part of water; insoluble in ethanol (95%), propan-2-ol, and common organic solvents. Specific surface area 0.70 m2 /g
Dextrates may be heated to 508C without any appreciable darkening of color. Dextrates should be stored in a well-closed container in conditions that do not exceed 258C and 60% relative humidity. When correctly stored in unopened containers, dextrates has a shelf-life of 3 years.
At high temperatures and humidities, dextrates may react with substances containing a primary amino group (Maillard reaction).(8,9) Also incompatible with oxidizing agents.
Dextrates is produced by controlled enzymatic hydrolysis of starch. The product is spray-crystallized, and may be dried to produce an anhydrous form.
Dextrates is used in oral pharmaceutical formulations and is generally regarded as a relatively nontoxic and nonirritant material.
Observe normal handling precautions appropriate to the circumstances and quantity of material handled. Eye protection, gloves, and a dust mask are recommended.
GRAS listed. Included in the FDA Inactive Ingredient Database (oral; tablets, chewable and sustained action). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Dextrose.
Only the hydrated form of dextrates is currently commercially available