

Name: Calcium Carbonate
CAS No: Carbonic acid, calcium salt (1 : 1) [471-34-1]
BP: Calcium Carbonate JP: Precipitated Calcium Carbonate PhEur: Calcium Carbonate USP: Calcium Carbonate
Calcii carbonas; calcium carbonate (1 : 1); carbonic acid calcium salt (1 : 1); creta preparada; Destab; E170; MagGran CC; Micromite; Pharma-Carb; precipitated carbonate of lime; precipitated chalk; Vitagran; Vivapress Ca; Witcarb.
Carbonic acid, calcium salt (1 : 1) [471-34-1]
CaCO3 100.09
See Section 4.
Calcium carbonate, employed as a pharmaceutical excipient, is mainly used in solid-dosage forms as a diluent.(1–4) It is also used as a base for medicated dental preparations,(5) as a buffering agent, and as a dissolution aid in dispersible tablets. Calcium carbonate is used as a bulking agent in tablet sugar-coating processes and as an opacifier in tablet film-coating. Calcium carbonate is also used as a food additive and therapeutically as an antacid and calcium supplement
Calcium carbonate occurs as an odorless and tasteless white powder or crystals.
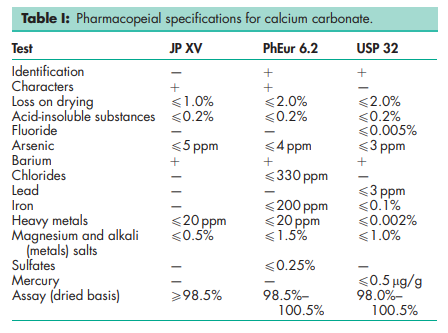
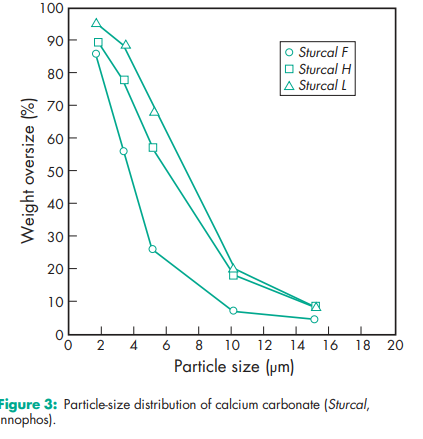
See Table I. See also Section 18
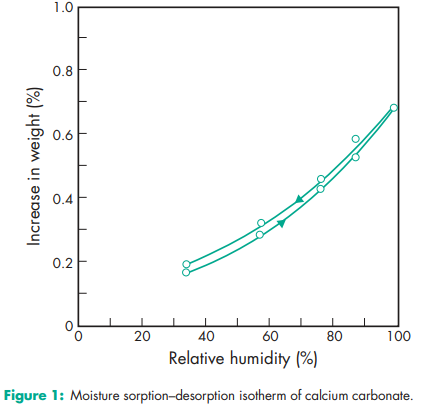
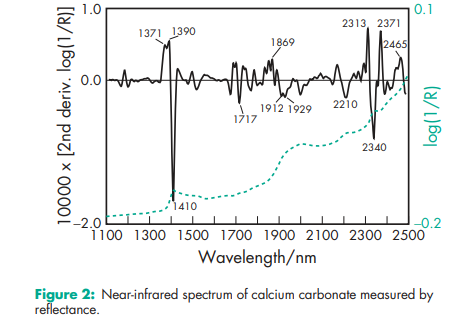
Acidity/alkalinity pH = 9.0 (10% w/v aqueous dispersion) Density (bulk) 0.8 g/cm3 Density (tapped) 1.2 g/cm3 Flowability Cohesive. Hardness (Mohs) 3.0 for Millicarb. Melting point Decomposes at 8258C. Moisture content see Figure 1. NIR spectra see Figure 2. Particle size see Figure 3. Refractive index 1.59 Solubility Practically insoluble in ethanol (95%) and water. Solubility in water is increased by the presence of ammonium salts or carbon dioxide. The presence of alkali hydroxides reduces solubility. Specific gravity 2.7 Specific surface area 6.21–6.47 m2 /g
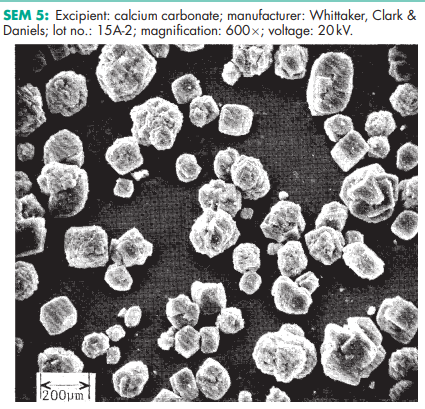
Calcium carbonate is stable and should be stored in a well-closed container in a cool, dry place. SEM 1: Excipient: calcium carbonate; manufacturer: Whittaker, Clark & Daniels; lot no.: 15A-3; magnification: 600; voltage: 20 kV. SEM 2: Excipient: calcium carbonate; manufacturer: Whittaker, Clark & Daniels; lot no.: 15A-3; magnification: 2400; voltage: 20 kV. SEM 3: Excipient: calcium carbonate; manufacturer: Whittaker, Clark & Daniels; lot no.: 15A-4; magnification: 600; voltage: 20 kV. SEM 4: Excipient: calcium carbonate; manufacturer: Whittaker, Clark & Daniels; lot no.: 15A-4; magnification: 2400; voltage: 20 kV. SEM 5: Excipient: calcium carbonate; manufacturer: Whittaker, Clark & Daniels; lot no.: 15A-2; magnification: 600; voltage: 20 kV. SEM 6: Excipient: calcium carbonate; manufacturer: Whittaker, Clark & Daniels; lot no.: 15A-2; magnification: 2400; voltage: 20 kV.





Incompatible with acids and ammonium salts (see also Sections 10 and 18).
Calcium carbonate is prepared by double decomposition of calcium chloride and sodium bicarbonate in aqueous solution. Density and fineness are governed by the concentrations of the solutions. Calcium carbonate is also obtained from the naturally occurring minerals aragonite, calcite, and vaterite.
Calcium carbonate is mainly used in oral pharmaceutical formulations and is generally regarded as a nontoxic material. However, calcium carbonate administered orally may cause constipation and flatulence. Consumption of large quantities (4–60 g daily) may also result in hypercalcemia or renal impairment.(6) Therapeutically, oral doses of up to about 1.5 g are employed as an antacid. In the treatment of hyperphosphatemia in patients with chronic renal failure, oral daily doses of 2.5–17 g have been used. Calcium carbonate may interfere with the absorption of other drugs from the gastrointestinal tract if administered concomitantly. LD50 (rat, oral): 6.45 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Calcium carbonate may be irritant to the eyes and on inhalation. Eye protection, gloves, and a dust mask are recommended. Calcium carbonate should be handled in a wellventilated environment. In the UK, the long-term (8-hour TWA) workplace exposure limit for calcium carbonate is 10 mg/m3 for total inhalable dust and 4 mg/m3 for respirable dust.(7)
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (buccal chewing gum, oral capsules and tablets; otic solutions; respiratory inhalation solutions). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Calcium carbonate is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. When calcium carbonate is used in tablets containing aspirin and related substances, traces of iron may cause discoloration. This may be overcome by inclusion of a suitable chelating agent. Grades with reduced lead levels are commercially available for use in antacids and calcium supplements. Directly compressible tablet diluents containing calcium carbonate and other excipients are commercially available. Examples of such grades are Barcroft CS90 (containing 10% starch), Barcroft CX50 (containing 50% sorbitol), and Barcroft CZ50 (containing 50% sucrose) available from SPI Pharma. Available from DMV International, are Cal-Carb 4450 PG (containing maltodextrin), and Cal-Carb 4457 and Cal-Carb 4462 (both containing pregelatinized corn starch). Two directly compressible grades containing only calcium carbonate are commercially available (Vivapress Ca 740 and Vivapress Ca 800, JRS Pharma). A specification for calcium carbonate is contained in the Food Chemicals Codex (FCC).(8) The EINECS number for calcium carbonate is 207-439-9. The PubChem Compound ID (CID) for calcium carbonate includes 10112 and 516889.