

Name: Calcium Hydroxide
CAS No: Calcium hydroxide [1305-62-0]
BP: Calcium Hydroxide JP: Calcium Hydroxide PhEur: Calcium Hydroxide USP: Calcium Hydroxide
Calcium hydrate; calcii hydroxidum; E526; hydrated lime; slaked lime
Calcium hydroxide [1305-62-0]
Ca(OH)2 74.1
See Section 4.
Calcium hydroxide is a strong alkali and is used as a pharmaceutical pH adjuster/buffer and antacid in topical medicinal ointments, creams, lotions, and suspensions, often as an aqueous solution (lime water).(1,2) It forms calcium soaps of fatty acids, which produce water-in-oil emulsions (calamine liniment), and it is also used as a topical astringent.(3,4) Calcium hydroxide is a common cosmetic ingredient in hairstraightening and hair-removal products, and in shaving preparations.(1) In dentistry, it is used as a filling agent and in dental pastes to encourage deposition of secondary dentine.(5) Calcium hydroxide was traditionally used as an escharotic in Vienna Paste.(6)
Calcium hydroxide occurs as a white or almost white, crystalline or granular powder. It has a bitter, alkaline taste. Calcium hydroxide readily absorbs carbon dioxide to form calcium carbonate.
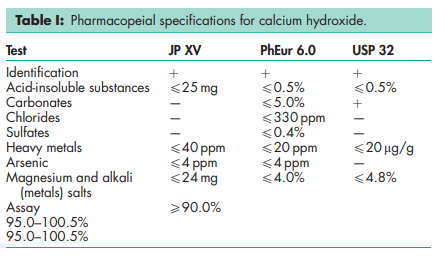
See Table I.
Acidity/alkalinity pH = 12.4 (saturated solution at 258C) Density 2.08–2.34 g/cm3 Melting point When heated above 5808C, it dehydrates forming the oxide. Solubility Soluble in glycerol and ammonium chloride solutions; dissolves in sucrose solutions to form calcium saccharosates;(2) soluble in acids with the evolution of heat; soluble 1 in 600 water (less soluble in hot water); insoluble in ethanol (95%).
Calcium hydroxide should be stored in an airtight container, in a cool, dry, well-ventilated place. Calcium hydroxide powder may be sterilized by heating for 1 hour at a temperature of at least 1608C.(2)
Incompatible with strong acids, maleic anhydride, phosphorus, nitroethane, nitromethane, nitroparaffins, and nitropropane. Calcium hydroxide can be corrosive to some metals.
Calcium hydroxide is manufactured by adding water to calcium oxide, a process called slaking.
Calcium hydroxide is used in oral and topical pharmaceutical formulations. It is mildly toxic by ingestion. In the pure state, calcium hydroxide is a severe skin, eye, and respiratory irritant, and it is corrosive, causing burns. Typical exposure limits are TVL 5 mg/m3 in air.(7) LD50 (mouse, oral): 7.3 g/kg LD50 (rat, oral): 7.34 g/kg(8)
Observe normal precautions appropriate to the circumstances and quantity of the material handled. Avoid contact with eyes, skin, and clothing. Avoid breathing the dust. Gloves, eye protection, respirator, and other protective clothing should be worn. In the USA, the OSHA permissable exposure limit is 15 mg/m3 for total dust and 5 mg/m3 respirable fraction for calcium hydroxide.(9
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (intravenous and subcutaneous injections; oral suspensions and tablets; topical emulsions and creams). Included in parenteral preparations licensed in the UK.
Potassium hydroxide; sodium hydroxide.
A specification for calcium hydroxide is contained in the Food Chemicals Codex (FCC).(10) The EINECS number for calcium hydroxide is 215-137-3. The PubChem Compound ID (CID) for calcium hydroxide is 14777.