

Name: Calcium Stearate
CAS No: Octadecanoic acid calcium salt [1592-23-0]
BP: Calcium Stearate JP: Calcium Stearate PhEur: Calcium Stearate USP-NF: Calcium Stearate
Calcii stearas; calcium distearate; calcium octadecanoate; Deasit PC; HyQual; Kemistab EC-F; octadecanoic acid, calcium salt; stearic acid, calcium salt; Synpro.
Octadecanoic acid calcium salt [1592-23-0]
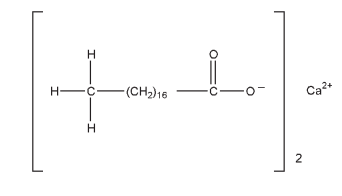
C36H70CaO4 607.03 (for pure material) The USP32–NF27 describes calcium stearate as a compound of calcium with a mixture of solid organic acids obtained from fats, and consists chiefly of variable proportions of calcium stearate and calcium palmitate. It contains the equivalent of 9.0–10.5% of calcium oxide. The PhEur 6.3 describes calcium stearate as a mixture of calcium salts of different fatty acids consisting mainly of stearic acid [(C17H35COO)2Ca] and palmitic acid [(C15H31COO)2Ca] with minor proportions of other fatty acids. It contains the equivalent of 9.0–10.5% of calcium oxide.
Calcium stearate is primarily used in pharmaceutical formulations as a lubricant in tablet and capsule manufacture at concentrations up to 1.0% w/w. Although it has good antiadherent and lubricant properties, calcium stearate has poor glidant properties. Calcium stearate is also employed as an emulsifier, stabilizing agent, and suspending agent, and is also used in cosmetics and food products.
Calcium stearate occurs as a fine, white to yellowish-white, bulky powder having a slight, characteristic odor. It is unctuous and free from grittiness.
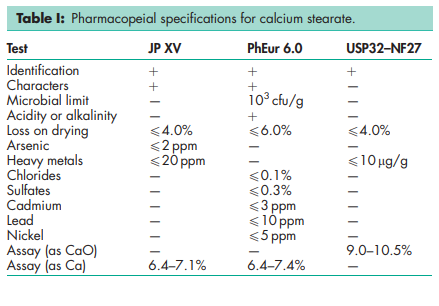
See Table I.
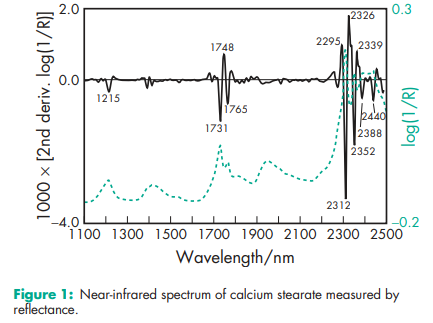
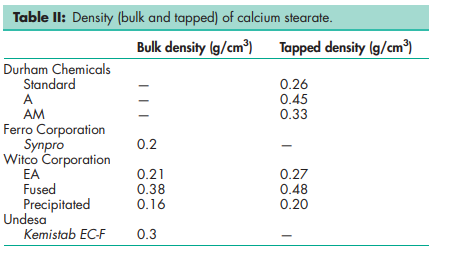
Acid value 191–203 Ash 9.9–10.3%; 9.2% for Synpro. Chloride <200 ppm Density (bulk and tapped) see Table II. Density (true) 1.064–1.096 g/cm3 ; 1.03 g/cm3 for Kemistab EC-F. Flowability 21.2–22.6% (Carr compressibility index) Free fatty acid 0.3–0.5%; 0.3% for Synpro. Melting point 149–1608C; 130–1568C for Kemistab EC-F; 1558C for Synpro. Moisture content 2.96%; 2.7% for Synpro. NIR spectra see Figure 1. Particle size distribution 1.7–60 mm; 100% through a 73.7 mm (#200 mesh); 99.5% through a 44.5 mm (#325 mesh). Shear strength 14.71 MPa Solubility Practically insoluble or insoluble in ethanol (95%), ether, chloroform, acetone, and water. Slightly soluble in hot alcohol, and hot vegetable and mineral oils. Soluble in hot pyridine.



Calcium stearate is stable and should be stored in a well-closed container in a cool, dry place.
Calcium stearate is prepared by the reaction of calcium chloride with a mixture of the sodium salts of stearic and palmitic acids. The calcium stearate formed is collected and washed with water to remove any sodium chloride.
Calcium stearate is used in oral pharmaceutical formulations and is generally regarded as a relatively nontoxic and nonirritant material.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Calcium stearate should be used in a well-ventilated environment; eye protection, gloves, and a respirator are recommended.
GRAS listed. Included in the FDA Inactive Ingredients Database (oral capsules and tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Magnesium stearate; stearic acid; zinc stearate.
Calcium stearate exhibits interesting properties when heated: softening between 120–1308C, and exhibiting a viscous consistency at approximately 1608C. At approximately 1008C, it loses about 3% of its weight, corresponding to 1 mole of water of crystallization. The crystalline structure changes at this point, leading to the collapse of the crystal lattice at a temperature of about 1258C.(1) Calcium stearate was studied as a component of a ‘cushioning pellet’ during compression of enteric-coated pellets to protect the enteric coating. The cushioning pellets were composed of stearic acid/microcrystalline cellulose (4 : 1 w/w) and were successful in avoiding rupture of the enteric coating during the compression process.(2) Calcium stearate was hot-melt extruded with testosterone in a study to characterize testosterone solid lipid microparticles to be applied as a transdermal delivery system. The results showed good release of the drug from the matrix.(3) See Magnesium stearate for further information and references. A specification for calcium stearate is contained in the Food Chemicals Codex (FCC).(4) The EINECS number for calcium stearate is 216-472-8. The PubChem Compound ID (CID) for calcium stearate is 15324.