

Name: AceticAcid,Glacial
CAS No: Ethanolicacid [64-19-7]
BP:GlacialAceticAcid JP:GlacialAceticAcid PhEur:AceticAcid,Glacial USP:GlacialAceticAcid
Acidum aceticum glaciale; E260; ethanoic acid; ethylic acid; methanecarboxylicacid;vinegaracid. SeealsoSections17and18.
Ethanolicacid [64-19-7]
C2H4O2 60.05
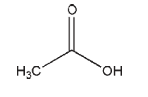
Glacial and diluted acetic acid solutions are widely used as acidifyingagents inavarietyofpharmaceutical formulationsand foodpreparations.Aceticacidisusedinpharmaceuticalproductsas abuffersystemwhencombinedwithanacetatesaltsuchassodium acetate.Aceticacidisalsoclaimedtohavesomeantibacterialand antifungalproperties.
Glacialaceticacidoccursasacrystallinemassoraclear,colorless volatilesolutionwithapungentodor
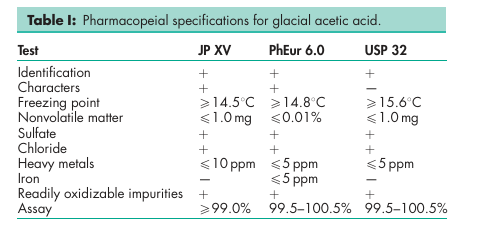
SeeTableI.
Acidity/alkalinity pH=2.4(1Maqueoussolution); pH=2.9(0.1Maqueoussolution); pH=3.4(0.01Maqueoussolution). Boilingpoint 1188C Dissociationconstant pKa=4.76 Flashpoint 398C(closedcup);578C(opencup). Meltingpoint 178C Refractiveindex nD 20=1.3718 Solubility Miscible with ethanol, ether, glycerin, water, and other fixed and volatile oils. Specific gravity 1.045
Acetic acid should be stored in an airtight container in a cool, dry place.
Acetic acid reacts with alkaline substances.
Acetic acid is usually made by one of three routes: acetaldehyde oxidation, involving direct air or oxygen oxidation of liquid acetaldehyde in the presence of manganese acetate, cobalt acetate, or copper acetate; liquid-phase oxidation of butane or naphtha; methanol carbonylation using a variety of techniques
Acetic acid is widely used in pharmaceutical applications primarily to adjust the pH of formulations and is thus generally regarded as relatively nontoxic and nonirritant. However, glacial acetic acid or solutions containing over 50% w/w acetic acid in water or organic solvents are considered corrosive and can cause damage to skin, eyes, nose, and mouth. If swallowed glacial acetic acid causes severe gastric irritation similar to that caused by hydrochloric acid.(1) Dilute acetic acid solutions containing up to 10% w/w of acetic acid have been used topically following jellyfish stings.(2) Dilute acetic acid solutions containing up to 5% w/w of acetic acid have also been applied topically to treat wounds and burns infected with Pseudomonas aeruginosa.(3) The lowest lethal oral dose of glacial acetic acid in humans is reported to be 1470mg/kg.(4) The lowest lethal concentration on inhalation in humans is reported to be 816ppm.(4) Humans, are, however, estimated to consume approximately 1g/day of acetic acid from the diet. LD50 (mouse, IV): 0.525g/kg(4) LD50 (rabbit, skin): 1.06g/kg LD50 (rat, oral): 3.31g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Acetic acid, particularly glacial acetic acid, can cause burns on contact with the skin, eyes, and mucous membranes. Splashes should be washed with copious quantities of water. Protective clothing, gloves, and eye protection are recom mended.
GRASlisted. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (injections, nasal, ophthalmic,and oral preparations). Included in parenteral and nonparenteral preparations licensed in the UK.
Acetic acid; artificial vinegar; dilute acetic acid. Acetic acid Comments A diluted solution of glacial acetic acid containing 30–37% w/w of acetic acid. See Section 18. Artificial vinegar Comments A solution containing 4% w/w of acetic acid. Dilute acetic acid Comments A weak solution of acetic acid which may contain between 6–10% w/w of acetic acid. See Section 18.
In addition to glacial acetic acid, many pharmacopeias contain monographs for diluted acetic acid solutions of various strengths. For example, the USP32–NF27 has a monograph for acetic acid, which is defined as an acetic acid solution containing 36.0–37.0% w/w of acetic acid. Similarly, the BP 2009 contains separate monographs for glacial acetic acid, acetic acid (33%), and acetic acid (6%). Acetic acid (33%) BP 2009 contains 32.5–33.5% w/w of acetic acid. Acetic acid (6%) BP 2009 contains 5.7–6.3% w/w of acetic acid. The JP XV also contains a monograph for acetic acid that specifies that it contains 30.0–32.0% w/w of acetic acid. A specification for glacial acetic acid is contained in the Food Chemicals Codex (FCC).(5) The EINECSnumberforacetic acid is 200-580-7. The PubChem Compound ID (CID) for glacial acetic acid is 176.