

Name: Carbon Dioxide
CAS No: Carbon dioxide [124-38-9]
BP: Carbon Dioxide JP: Carbon Dioxide PhEur: Carbon Dioxide USP: Carbon Dioxide
Carbonei dioxidum; carbonic acid gas; carbonic anhydride; E290
Carbon dioxide [124-38-9]
CO2 44.01
See Section 4
Carbon dioxide and other compressed gases such as nitrogen and nitrous oxide are used as propellants for topical pharmaceutical aerosols. They are also used in other aerosol products that work satisfactorily with the coarse aerosol spray that is produced with compressed gases, e.g. cosmetics, furniture polish, and window cleaners.(1–3) The advantages of compressed gases as aerosol propellants are that they are less expensive; are of low toxicity; and are practically odorless and tasteless. Also, in comparison to liquefied gases, their pressures change relatively little with temperature. However, the disadvantages of compressed gases are that there is no reservoir of propellant in the aerosol and pressure consequently decreases as the product is used. This results in a change in spray characteristics. Additionally, if a product that contains a compressed gas as a propellant is actuated in an inverted position, the vapor phase, rather than the liquid phase, is discharged. Most of the propellant is contained in the vapor phase and therefore some of the propellant will be lost and the spray characteristics will be altered. Also, sprays produced using compressed gases are very wet. Valves, such as the vapor tap or double dip tube, are currently available and will overcome these problems. Carbon dioxide is also used to displace air from pharmaceutical products by sparging and hence to inhibit oxidation. As a food additive it is used to carbonate beverages and to preserve foods such as bread from spoilage by mold formation, the gas being injected into the space between the product and its packaging.(4,5) Solid carbon dioxide is also widely used to refrigerate products temporarily, while liquid carbon dioxide, which can be handled at temperatures up to 318C under high pressure, is used as a solvent for flavors and fragrances, primarily in the perfumery and food manufacturing industries
Carbon dioxide occurs naturally as approximately 0.03% v/v of the atmosphere. It is a colorless, odorless, noncombustible gas with a faint acid taste. Solid carbon dioxide, also known as dry ice, is usually encountered as white-colored pellets or blocks.
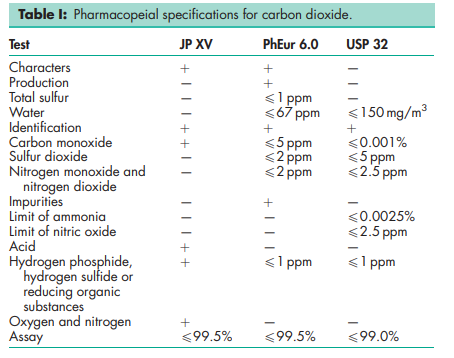
See Table I.
Density 0.714 g/cm3 for liquid at 258C; 0.742 g/cm3 for vapor at 258C. Flammability Nonflammable. Solubility 1 in about 1 of water by volume at normal temperature and pressure. Vapor density (absolute) 1.964 g/m3 Vapor density (relative) 1.53 (air = 1) Viscosity (kinematic) 0.14 mm2 /s (0.14 cSt) at 17.88C.
Extremely stable and chemically nonreactive. Store in a tightly sealed cylinder. Avoid exposure to excessive heat.
Carbon dioxide is generally compatible with most materials although it may react violently with various metal oxides or reducing metals such as aluminum, magnesium, titanium, and zirconium. Mixtures with sodium and potassium will explode if shocked
Carbon dioxide is obtained industrially in large quantities as a byproduct in the manufacture of lime; by the incineration of coke or other carbonaceous material; and by the fermentation of glucose by yeast. In the laboratory it may be prepared by dropping acid on a carbonate.
In formulations, carbon dioxide is generally regarded as an essentially nontoxic material.
Handle in accordance with standard procedures for handling metal cylinders containing liquefied or compressed gases. Carbon dioxide is an asphyxiant, and inhalation in large quantities is hazardous. It should therefore be handled in a well-ventilated environment equipped with suitable safety devices for monitoring vapor concentration. It should be noted that carbon dioxide is classified as a greenhouse gas responsible for global warming. At the present time there are no restrictions on its use for aerosols and other pharmaceutical applications. In the UK, the workplace exposure limits for carbon dioxide are 9150 mg/m3 (5000 ppm) long-term (8-hour TWA) and 27 400 mg/m3 (15 000 ppm) short-term (15-minute).(6) In the USA, the permissible exposure limits are 9000 mg/m3 (5000 ppm) longterm and the recommended exposure limits are 18 000 mg/m3 (10 000 ppm) short-term and 54 000 mg/m3 (30 000 ppm) maximum, short-term.(7) Solid carbon dioxide can produce severe burns in contact with the skin and appropriate precautions, depending on the circumstances and quantity of material handled, should be taken. A face shield and protective clothing, including thick gloves, are recommended
GRAS listed. Accepted for use in Europe as a food additive. Included in the FDA Inactive Ingredients Database (aerosol formulation for nasal preparations; IM and IV injections). Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Nitrogen; nitrous oxide.
Supercritical carbon dioxide has been used in the formation of fine powders of stable protein formulations.(8,9) Carbon dioxide has also been investigated for its suitability in Aerosol Solvent Extraction Systems (ASES), to generate microparticles of proteins suitable for aerosol delivery from aqueous based solutions.(10) A specification for carbon dioxide is contained in the Food Chemicals Codex (FCC).(11) The EINECS number for carbon dioxide is 204-696-9. The PubChem Compound ID (CID) for carbon dioxide is 280.