

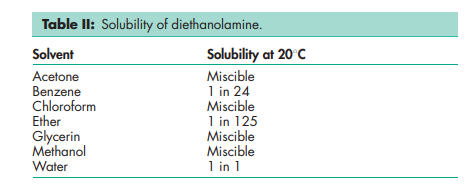
Name: Diethanolamine
CAS No: 2,20-Iminobisethanol [111-42-2]
USP-NF: Diethanolamine
Bis(hydroxyethyl)amine; DEA; diethylolamine; 2,20-dihydroxydiethylamine; diolamine; 2,20-iminodiethanol.
2,20-Iminobisethanol [111-42-2]
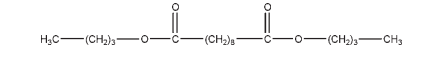
C4H11NO2 105.14
Diethanolamine is primarily used in pharmaceutical formulations as a buffering agent, such as in the preparation of emulsions with fatty acids. In cosmetics and pharmaceuticals it is used as a pH adjuster and dispersant. Diethanolamine has also been used to form the soluble salts of active compounds, such as iodinated organic acids that are used as contrast media. As a stabilizing agent, diethanolamine prevents the discoloration of aqueous formulations containing hexamethylenetetramine-1,3-dichloropropene salts. Diethanolamine is also used in cosmetics.
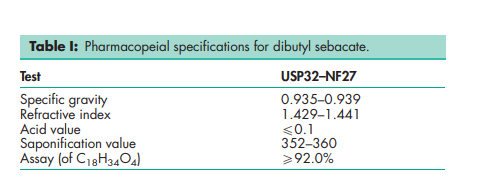
The USP32–NF27 describes diethanolamine as a mixture of ethanolamines consisting largely of diethanolamine. At about room temperature it is a white, deliquescent solid. Above room temperature diethanolamine is a clear, viscous liquid with a mildly ammoniacal odor
See Table I
Acidity/alkalinity pH = 11.0 for a 0.1 N aqueous solution. Autoignition temperature 6628C Boiling point 268.88C Density 1.0881 g/cm3 at 308C; 1.0693 g/cm3 at 608C. Dissociation constant pKa = 8.88 Flash point 1388C (open cup) Hygroscopicity Very hygroscopic. Melting point 288C Refractive index n D 30 = 1.4753 Surface tension 49.0 mN/m (49.0 dynes/cm) at 208C Vapor density (relative) 3.65 (air = 1) Vapor pressure >1 Pa at 208C Viscosity (dynamic) 351.9 mPa s (351.9 cP) at 308C; 53.85 mPa s (53.85 cP) at 608C.
Diethanolamine is hygroscopic and light- and oxygen-sensitive; it should be stored in an airtight container, protected from light, in a cool, dry place.
Diethanolamine is a secondary amine that contains two hydroxy groups. It is capable of undergoing reactions typical of secondary amines and alcohols. The amine group usually exhibits the greater activity whenever it is possible for a reaction to take place at either the amine or a hydroxy group. Diethanolamine will react with acids, acid anhydrides, acid chlorides, and esters to form amide derivatives, and with propylene carbonate or other cyclic carbonates to give the corresponding carbonates. As a secondary amine, diethanolamine reacts with aldehydes and ketones to yield aldimines and ketimines. Diethanolamine also reacts with copper to form complex salts. Discoloration and precipitation will take place in the presence of salts of heavy metals.
Diethanolamine is prepared commercially by the ammonolysis of ethylene oxide. The reaction yields a mixture of monoethanolamine, diethanolamine, and triethanolamine which is separated to obtain the pure products.
Diethanolamine is used in topical and parenteral pharmaceutical formulations, with up to 1.5% w/v being used in intravenous infusions. Experimental studies in dogs have shown that intravenous administration of larger doses of diethanolamine results in sedation, coma, and death. Animal toxicity studies suggest that diethanolamine is less toxic than monoethanolamine, although in rats the oral acute and subacute toxicity is greater.(1) Diethanolamine is said to be heptacarcinogenic in mice and has also been reported to induce hepatic choline deficiency in mice.(2) Diethanolamine is an irritant to the skin, eyes, and mucous membranes when used undiluted or in high concentration. However, in rabbits, aqueous solutions containing 10% w/v diethanolamine produce minor irritation. The lethal human oral dose of diethanolamine is estimated to be 5–15 g/kg body-weight. The US Cosmetic Ingredient Review Expert Panel evaluated diethanolamine and concluded that it is safe for use in cosmetic formulations designed for discontinuous, brief use followed by thorough rinsing from the surface of the skin. In products intended for prolonged contact with the skin, the concentration of ethanolamines should not exceed 5%. Diethanolamine should not be used in products containing N-nitrosating agents.(1) See also Section 18. LD50 (guinea pig, oral): 2.0 g/kg(3) LD50 (mouse, IP): 2.3 g/kg LD50 (mouse, oral): 3.3 g/kg LD50 (rabbit, skin): 12.2 g/kg LD50 (rat, IM): 1.5 g/kg LD50 (rat, IP): 0.12 g/kg LD50 (rat, IV): 0.78 g/kg LD50 (rat, oral): 0.71 g/kg LD50 (rat, SC): 2.2 g/kg
Diethanolamine is irritating to the skin, eyes, and mucous membranes. Protective clothing, gloves, eye protection, and a respirator are recommended. Ideally, diethanolamine should be handled in a fume cupboard.(4) Diethanolamine poses a slight fire hazard when exposed to heat or flame.
Included in the FDA Inactive Ingredients Database (IV infusions, ophthalmic solutions, and topical preparations). Included in medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Monoethanolamine; triethanolamine.
Through a standard battery of rodent studies, diethanolamine has been identified by the US National Toxicology Program as a potential carcinogen following topical administration. Several possible confounding issues have been noted during the review of these studies, which may affect the ultimate conclusion made regarding the carcinogenicity of diethanolamine and the relevance of these findings to humans. Diethanolamine is not permitted for use in cosmetics sold within the EU. The EINECS number for diethanolamine is 203-868-0. The PubChem Compound ID (CID) for diethanolamine is 8113.