

Name: Carrageenan
CAS No: Carrageenan [9000-07-1], i-Carrageenan [9062-07-1], k-Carrageenan [11114-20-8], l-Carrageenan [9064-57-7],
USP-NF: Carrageenan
Chondrus extract; E407; Gelcarin; Genu; Grindsted; Hygum TP-1; Irish moss extract; Marine Colloids; SeaSpen PF; Viscarin.
Carrageenan [9000-07-1], i-Carrageenan [9062-07-1], k-Carrageenan [11114-20-8], l-Carrageenan [9064-57-7],
The USP32–NF27 describes carrageenan as the hydrocolloid obtained by extraction with water or aqueous alkali from some members of the class Rhodophyceae (red seaweed). It consists chiefly of potassium, sodium, calcium, magnesium, and ammonium sulfate esters of galactose and 3,6-anhydrogalactose copolymers. These hexoses are alternately linked at the a-1,3 and b-1,4 sites in the polymer.
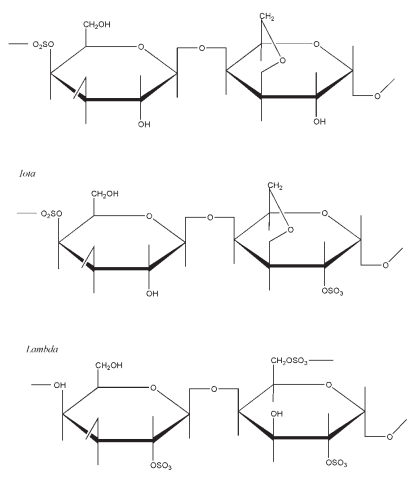
The carrageenans are divided into three families according to the position of sulfate groups and the presence or absence of anhydrogalactose. l-Carrageenan (lambda-carrageenan) is a nongelling polymer containing about 35% ester sulfate by weight and no 3,6- anhydrogalactose. i-Carrageenan (iota-carrageenan) is a gelling polymer containing about 32% ester sulfate by weight and approximately 30% 3,6- anhydrogalactose. k-Carrageenan (kappa-carrageenan) is a strongly gelling polymer which has a helical tertiary structure that allows gelling.(1) It contains 25% ester sulfate by weight and approximately 34% 3,6- anhydrogalactose.
Carrageenan is used in a variety of nonparenteral dosage forms, including suspensions (wet and reconstitutable), emulsions, gels, creams, lotions, eye drops, suppositories, tablets, and capsules. In suspension formulations, usually only the i-carrageenen and lcarrageenan fractions are used. l-Carrageenan is generally used at levels of 0.7% w/v or less, and provides viscosity to the liquid. Carrageenan has been shown to mask the chalkiness of antacid suspensions when used as a suspending agent in these preparations.(2) When used in concentrations of 0.1–0.5%, carrageenan gives stable emulsions. Carrageenan is used in hand lotions and creams to provide slip and improved ‘rub out’. i-Carrageenan develops a shear-thinning thixotropic gel, which can be easily poured after shaking. When i-carrageenan is used, the presence of calcium ions is required for the gel network to become established. With pure i-carrageenan, about 0.4% w/v is required for most suspensions plus the addition of calcium. However, if SeaSpen PF is used, it must be at about 0.75% w/v level, although no additional calcium is required as this is already present in the product to control the rate of gelation. Studies on the effect of carrageenan and other colloids on mucoadhesion of drugs to the oropharyngeal areas(3,4) have shown that carrageenan had the greatest propensity for adhesion and can be used in formulations for oral and buccal drug delivery. The application of carrageenan in topical gel bases has been examined,(5–7) and the findings indicate that the use of carrageenan in these dosage forms is most likely to be dependent on the active drug, owing to the potential for ionic interactions. In the case of topical gels, a combination of i, k-, and lcarrageenans produces a spreadable gel with acceptable tactile sensation, resulting in drug release that is more likely to follow diffusion kinetics. Incorporation of carrageenan into tablet matrices with various drugs and other excipients to alter release profiles has been studied, illustrating that the carrageenans have good tablet-binding properties.(8–12) Furthermore, the inclusion of calcium or potassium salts into the tablet creates a microenvironment for gelation to occur, which further controls drug release. There have also been several references to the use of carrageenan in chewable tablets having a confectionary texture.(13,14) This approach to creating a novel dosage form requires the use of both icarrageenan and k-carrageenan, to prevent moisture loss and texture changes that occur over time. See also Section 10. Carrageenan has been used for the microencapsulation of proteins(15) and probiotic bacteria.(16) Hydrogels have also been prepared by crosslinking with gelatin and k-carrageenan for oral delivery of probiotic bacteria.(17) It has also been used as beads in the preparation of controlled release systems.(18,19) Hydrogel beads based on k-carrageenan and sodium alginate/chitosan are being used as new carriers for drug loading and controlled delivery systems.(20,21) k-Carrageenan is known as a novel pelletization aid in the manufacture of pellets by extrusion/spheronization and has the best pelletization behavior.(22–25) l-Carrageenan is also able to nanoencapsulate drug molecules spontaneously, hence controling drug release.(26) The presence of carrageenan induces the formation of smaller particles compared to those formed in the absence of polymer, and their average size depends on the nature and concentration of the polysaccharide used.(27) Studies have shown that carrageenan compounds block infections by the herpes simplex virus;(28) human cytomegalovirus; human papilloma virus;(29) Sindbis virus; vesicular stomatitis virus; and HIV.(30) A combined k- and l-carrageenan formulation is currently being investigated as the active ingredient in a topical microbicide used to prevent the sexual transmission of HIV.(31–33) In combination with chitosan, agar and polyvinyl pyrrolidone, carrageenan forms a water-insoluble complex which is able to absorb large amounts of body fluids, and is used as an effective wound dressing.(34–36) Carrageenan is used in the preparation of hard and soft capsule shells.(37) It is also used in toothpastes and cosmetic preparations such as conditioners and shampoos.(38,3
Carrageenan, when extracted from the appropriate seaweed source, is a yellow-brown to white colored, coarse to fine powder that is odorless and tasteless.
See Table I
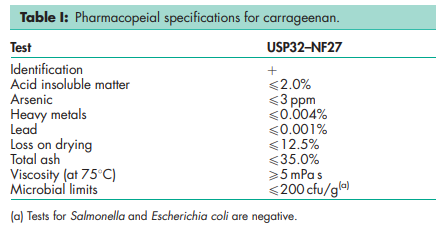
Because of the vast differences in the material that can be referred to as carrageenan, it is difficult to give descriptions of typical properties. See Table II. NIR spectra see Figures 1, 2, 3, and 4. Solubility Soluble in water at 808C. See Tables II and III. Viscosity (dynamic) 5 mPa s (5 cP) at 758C. See Table II.
Carrageenan is a stable, though hygroscopic, polysaccharide and should be stored in a cool, dry place. Carrageenan in solution has maximum stability at pH 9 and should not be heat processed at pH values below 3.5. Acid and oxidizing agents may hydrolyze carrageenan in solution leading to loss of physical properties through cleavage of glycosidic bonds. Acid hydrolysis depends on pH, temperature and time. The acid hydrolysis takes place only when the carrageenan is dissolved, and the hydrolysis is accelerated as the processing temperature and/or the processing time is increased. However, when the carrageenan is in its gelled state the acid hydrolysis no longer takes place; see Table IV.
Carrageenan can react with cationic materials. If complexation of cationic materials, with associated modification of the active compound’s solubility, is undesirable, the use of carrageenan is not recommended. Carrageenan may interact with other charged macromolecules, e.g. proteins, to give various effects such as viscosity increase, gel formation, stabilization or precipitation.
The main species of seaweed from which carrageenan is manufactured are Eucheuma, Chondrus, and Gigartina. The weed is dried quickly to prevent degradation, and is then baled for shipment to processing facilities. The seaweed is repeatedly washed to remove gross impurities such as sand, salt, and marine life, and then undergoes a hot alkali extraction process, releasing the carrageenan from the cell. Once it is in a hot solution, carrageenan undergoes clarification and concentration in solution and is converted to powder. Three processes can be used to remove the carrageenan from solution. The first is a ‘freeze–thaw’ technique. The solution is gelled with various salts, then the gels are frozen. Upon thawing, the water is removed and the resultant mass, primarily carrageenan and salt, is ground to the desired particle size. The second method, referred to as the ‘alcohol precipitation method’ takes the concentrated solution of carrageenan and places it in alcohol. This causes the carrageenan to precipitate out of solution. The cosolvents are evaporated and the precipitated carrageenan is dried and ground to the desired particle size. The third method is the ‘KCl precipitation’ process, where after hot extraction, the filtrate is evaporated to reduce the filtrate volume. The filtrate is then extruded through spinnerets into a cold 1.0–1.5% solution of potassium chloride. The resulting gel threads are washed with KCl solution and are pressed, dried and milled to carrageenan powder.(2) Commercial carrageenan is usually standardized by blending different batches of carrageenan and adding sugar or salt to obtain the desired gelling or thickening properties.(40)
Carrageenan is widely used in numerous food applications and is increasingly being used in pharmaceutical formulations. Carrageenan is generally regarded as a relatively nontoxic and nonirritating material when used in nonparenteral pharmaceutical formulations. However, carrageenan is known to induce inflammatory responses in laboratory animals, and for this reason it is frequently used in experiments for the investigation of anti-inflammatory drugs.(41–45) Animal studies suggest that degraded carrageenan (which is not approved for use in food products) may be associated with cancer in the intestinal tract, although comparable evidence does not exist in humans.(46) The WHO has set an acceptable daily intake of carrageenan of ‘not specified’ as the total daily intake was not considered to represent a hazard to health.(47) In the UK, the Food Advisory Committee has recommended that carrageenan should not be used as an additive for infant formulas.(48) LD50 (rat, oral): >5 g/kg LD50 (rabbit, skin): >2 g/kg/4 h LC50 (rat, inhalation): >0.93 mg/L(49)
Observe normal precautions appropriate to the circumstances and quantity of material handled
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (dental; oral capsules, granules, powders and syrups; topical; transdermal preparations; and controlled-release film preparations). Included in the Canadian List of Acceptable Non-medicinal Ingredients. Included in nonparenteral medicines (oral granules, capsules (shells), and orodispersible tablets) licensed in the UK.
A specification for carrageenan is included in the Japanese Pharmaceutical Excipients (JPE).(50) The EINECS number for carrageenan is 232-524-2.