

Name: Dimethyl Ether
CAS No: Methoxymethane [115-10-6]
None adopted
Dimethyl oxide; DME; Dymel A; methoxymethane; methyl ether; oxybismethane; wood ether.
Methoxymethane [115-10-6]
C2H6O 46.07
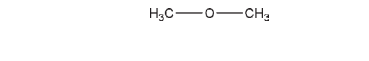
Dimethyl ether may be used as an aerosol propellant for topical aerosol formulations in combination with hydrocarbons and other propellants.(1–4) Generally, it cannot be used alone as a propellant owing to its high vapor pressure. Dimethyl ether is a good solvent and has the unique property of high water solubility, compared to other propellants. It has frequently been used with aqueous aerosols. A coarse, wet, spray is formed when dimethyl ether is used as a propellant. Dimethyl ether is also used as a propellant in cosmetics such as hair sprays, and in other aerosol products such as air fresheners and fly sprays. Dimethyl ether is additionally used as a refrigerant.
Dimethyl ether is a liquefied gas and exists as a liquid at room temperature when contained under its own vapor pressure, or as a gas when exposed to room temperature and pressure. It is a clear, colorless, virtually odorless liquid. In high concentrations, the gas has a faint ether-like odor.
—
Autoignition temperature 3508C Boiling point 23.68C Critical temperature 126.98C Density 0.66 g/cm3 for liquid at 258C. Flammability The pure material is flammable; limit of flammability is 3.4–18.2% v/v in air. Aqueous mixtures are nonflammable. Freezing point 138.58C Flash point 418C Heat of combustion 28.9 kJ/g (6900 cal/g) Kauri-butanol value 60 Solubility Soluble in acetone, chloroform, ethanol (95%), ether, and 1 in 3 parts of water. Dimethyl ether is generally miscible with water, nonpolar materials, and some semipolar materials. For pharmaceutical aerosols, ethanol (95%) is the most useful cosolvent. Glycols, oils, and other similar materials exhibit varying degrees of miscibility with dimethyl ether. Surface tension 16 mN/m (16 dynes/cm) at –108C Vapor density (absolute) 2.058 g/m3 at standard temperature and pressure. Vapor density (relative) 1.596 (air = 1) Vapor pressure 592 kPa at 258C (63 psig at 21.18C); 1301 kPa at 548C.
The liquefied gas is stable when used as a propellant. However, exposure to the air for long periods of time may result in explosive peroxides being slowly formed. Solutions of liquid dimethyl ether should not be concentrated either by distillation or by evaporation. Dimethyl ether should be stored in tightly closed metal cylinders in a cool, dry place.
Dimethyl ether is an aggressive solvent and may affect the gasket materials used in aerosol packaging. Oxidizing agents, acetic acid, organic acids, and anhydrides should not be used with dimethyl ether.
Dimethyl ether is prepared by the reaction of bituminous or lignite coals with steam in the presence of a finely divided nickel catalyst. This reaction produces formaldehyde, which is then reduced to methanol and dimethyl ether. Dimethyl ether may also be prepared by the dehydration of methanol.
Dimethyl ether may be used as a propellant and solvent in topical pharmaceutical aerosols, and is generally regarded as an essentially nontoxic and nonirritant material when used in such applications. However, inhalation of high concentrations of dimethyl ether vapor is harmful. Additionally, skin contact with dimethyl ether liquid may result in freezing of the skin and severe frostbite. When used in topical formulations, dimethyl ether may exert a chilling effect on the skin, although if it is used as directed the propellant quickly vaporizes and is nonirritating. LD50 (mouse, inhalation): 386 000 ppm/30 min(5) LD50 (rat, inhalation): 308 g/m3
Dimethyl ether is usually encountered as a liquefied gas, and appropriate precautions for handling such materials should be taken. Eye protection, gloves, and protective clothing are recommended. Dimethyl ether should be handled in a well-ventilated environment. Dimethyl ether vapor is heavier than air and does not support life; therefore, when cleaning large tanks that have contained this material, adequate provisions for oxygen supply in the tanks must be made in order to protect workers cleaning the tanks. In the UK, the long-term (8-hour TWA) exposure limit for dimethyl ether is 766 mg/m3 (400 ppm). The short-term (15-minute) exposure limit is 958 mg/m3 (500 ppm).(6)
Included in the FDA Inactive Ingredients Database (topical aerosols). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Hydrocarbons (HC).
Since the solubility of dimethyl ether in water is about 35%, it can be used to good effect in aqueous aerosol products. It also has antimicrobial effects that are organism-dependent.(7) The EINECS number for dimethyl ether is 204-065-8. The PubChem Compound ID (CID) for dimethyl ether is 8254.