

Name: Dimethyl Sulfoxide
CAS No: Sulfinylbismethane [67-68-5]
BP: Dimethyl Sulfoxide PhEur: Dimethyl Sulfoxide USP: Dimethyl Sulfoxide
Deltan; dimexide; dimethylis sulfoxidum; dimethyl sulphoxide; DMSO; Kemsol; methylsulfoxide; Procipient; Rimso-50; sulphinylbismethane
Sulfinylbismethane [67-68-5]
C2H6OS 78.13
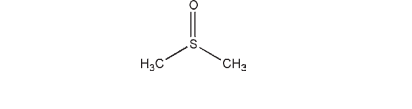
Dimethyl sulfoxide is a highly polar substance that is aprotic, therefore lacking acidic and basic properties. It has exceptional solvent properties for both organic and inorganic components, which are derived from its capacity to associate with both ionic species and neutral molecules that are either polar or polarizable. Dimethyl sulfoxide enhances the topical penetration of drugs owing to its ability to displace bound water from the stratum corneum; this is accompanied by the extraction of lipids and configurational changes of proteins.(1) The molecular interactions between dimethyl sulfoxide and the stratum corneum, as a function of depth and time, have been described.(2) Much of the enhancement capacity is lost if the solvent is diluted. Increases in drug penetration have been reported with dimethyl sulfoxide concentrations as low as 15%, but significant increases in permeability generally require concentrations higher than 60–80%. Furthermore, while low molecular weight substances can penetrate quickly into the deep layers of the skin, the appreciable transport of molecules with a molecular weight of more than 3000 is difficult. Dimethyl sulfoxide is now incorporated into a number of regulated products for healthcare and drug delivery applications, including stabilizing product formulations, sustained-release applications, and for the delivery of medical polymers.(3) The use of dimethyl sulfoxide to improve transdermal delivery has been reported for diclofenac,(4,5) ciclosporin,(6) timolol,(7) and a wide range of other drugs.(8,9) Dimethyl sulfoxide has also been used in the formulation of an injection containing allopurinol.(10) It has also been investigated for use in an experimental parenteral preparation for the treatment of liver tumors.(11) In paint formulations of idoxuridine, dimethyl sulfoxide acts both as a solvent to increase drug solubility and a means of enabling penetration of the antiviral agent to the deeper levels of the epidermis. See Table I. Dimethyl sulfoxide has also been investigated as a potential therapeutic agent in conditions such as scleroderma, interstitial cystitis,(12) rheumatoid arthritis, and acute musculoskeletal injuries, and as an analgesic.(13–17) It has also been recommended for the treatment of anthracycline extravasation(18–21) and has been investigated as a potential cryoprotectant.(22,23)
Dimethyl sulfoxide occurs as a colorless, viscous liquid, or as colorless crystals that are miscible with water, alcohol, and ether. The material has a slightly bitter taste with a sweet aftertaste, and is odorless, or has a slight odor characteristic of dimethyl sulfoxide. Dimethyl sulfoxide is extremely hygroscopic, absorbing up to 70% of its own weight in water with evolution of heat.
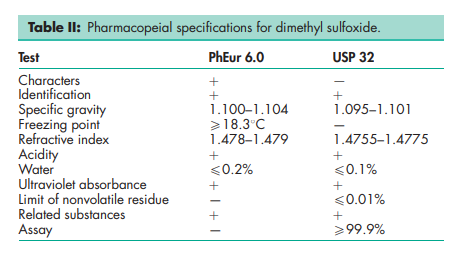
See Table II.
Acidity/alkalinity pH = 8.5 (for a 50 : 50 mixture with water) Autoignition temperature 2158C Boiling point 1898C Density 1.0955 g/cm3 at 258C for Procipient Dielectric constant 48.9 at 208C Dipole moment (D) 4.3 at 208C(24) Dissociation constant pKa = 31.3(24) Enthalpy of fusion 3.43 cal/mol(24) Enthalpy of vaporization 12.64 cal/mol at 258C(24) Flash point 958C (open cup) Partition coefficient log (octanol/water) = 2.03 Specific heat 0.7 cal/g (liquid) Solubility Miscible with water with evolution of heat; also miscible with ethanol (95%), ether and most organic solvents; immiscible with paraffins, hydrocarbons. Practically insoluble in acetone, chloroform, ethanol (95%), and ether. Vapor pressure 0.37 mm at 208C Viscosity (dynamic) 1.1 mPa s (1.1 cP) at 278C; 2.0 mPa s (2.0 cP) at 258C for Procipient; 2.47 mPa s (2.47 cP) at 208C.
Dimethyl sulfoxide is reasonably stable to heat, but upon prolonged reflux it decomposes slightly to methyl mercaptan and bismethylthiomethane. This decomposition is aided by acids, and is retarded by many bases. When heated to decomposition, toxic fumes are emitted. At temperatures between 40 and 608C, it has been reported that dimethyl sulfoxide suffers a partial breakdown, which is indicated by changes in physical properties such as refractive index, density, and viscosity.(25) Dimethyl sulfoxide should be stored in airtight, light-resistant containers. The PhEur 6.0 states that glass containers should be used. Contact with plastics should be avoided.
Dimethyl sulfoxide can react with oxidizing materials
Dimethyl sulfoxide is prepared by air oxidation of dimethyl sulfide in the presence of nitrogen oxides. It can also be obtained as a by product of wood pulp manufacture for the paper and allied industries.
Dimethyl sulfoxide has low systemic toxicity but causes local toxic effects.(26–28) It is readily absorbed after injection or after oral or percutaneous administration and is widely distributed throughout the body. Dimethyl sulfoxide acts as a primary irritant on skin, causing redness, burning, itching, and scaling; it also causes urticaria. Systemic symptoms include nausea, vomiting, chills, cramps, and lethargy; dimethyl sulfoxide can also cause increases in intraocular pressure. Administration of dimethyl sulfoxide by any route is followed by a garlic-like odor on the breath. Intravascular hemolysis and biochemical changes(29) and reversible neurological deterioration(30) have been reported following intravenous administration; however, it has been questioned whether these findings were directly attributable to dimethyl sulfoxide rather than to concomitant drug therapy or contaminants.(31) One report describes massive intracranial hemorrhage associated with ingestion of dimethyl sulfoxide.(32) Recently, a hypersensitivity reaction attributed to dimethyl sulfoxide has been reported.(33) In 1965, the FDA banned investigation in humans of dimethyl sulfoxide owing to the appearance of changes in the refractive index of the lens of the eye in experimental animals. However, in 1966, the FDA allowed the study of dimethyl sulfoxide in serious conditions such as scleroderma, persistent herpes zoster, and severe rheumatoid arthritis, and in 1968 permitted studies using short-term topical application of the solvent. By 1980, the FDA no longer specifically regulated investigations of dimethyl sulfoxide.(14) Dimethyl sulfoxide enhances the skin penetration of several drugs, which may result in producing the adverse effects associated with those drugs. LD50 (dog, IV): 2.5 g/kg(34) LD50 (rat, IP): 8.2 g/kg LD50 (rat, IV): 5.3 g/kg LD50 (rat, oral): 14.5 g/kg LD50 (rat, SC): 12 g/kg LD50 (mouse, IP): 2.5 g/kg LD50 (mouse, IV): 3.8 g/kg LD50 (mouse, oral): 7.9 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Dimethyl sulfoxide may cause irritation to the skin. Gloves and eye protection are recommended.
Included in the FDA Inactive Ingredients Database (IV infusions, SC implants, and topical preparations). Available in the USA as a 50% solution for irrigation in the treatment of interstitial cystitis. Also available in Canada as a 70% solution for use as a topical antifibrotic, and in Germany as a topical gel containing 10% dimethyl sulfoxide for the treatment of musculoskeletal and joint disorders. Included in topical formulations of idoxuridine and diclofenac licensed in the UK.
—
A 2.16% dimethyl sulfoxide solution in water is iso-osmotic with serum. Dimethyl sulfoxide has been used as a 50% aqueous solution for instillation into the bladder in the treatment of interstitial cystitis; it has also been tried clinically for a wide range of indications, including cutaneous and musculoskeletal disorders, but with little evidence of beneficial effects. Dimethyl sulfoxide has been shown to have bactericidal,(35) bacteriostatic,(35,36) and fungistatic(36) activity, although the concentration required is dependent on the organism present. The EINECS number for dimethyl sulfoxide is 200-664-3. The PubChem Compound ID (CID) for dimethyl sulfoxide is 679.